Snmmi Applauds Fda Approval Of New Metastatic Prostate Cancer Treatment
Reston, VAWith todays U.S. Food and Drug Administration approval of 177Lu-PSMA-617a radiopharmaceutical therapy to treat metastatic castration-resistant prostate cancer the Society of Nuclear Medicine and Molecular Imaging celebrates one of the greatest success stories in nuclear medicine history. The therapy, developed after years of extensive research, has been shown to reduce the risk of death by 38 percent and reduce the risk of progression by 60 percent in mCRPC patients treated with 177Lu-PSMA-617.
The new therapy depends on the use of PET scans to identify and treat patients whose metastatic prostate cancers express PSMA . We are delighted by the FDA approval of this transformational, innovative therapy for men with advanced metastatic castrate resistant prostate cancer, said SNMMI president Richard L. Wahl, MD, FACNM. We are proud of the society members who contributed substantially to this new theranostic paradigm, as well as all of the authors who published articles on this therapy in The Journal of Nuclear Medicine.
The FDA granted Priority Review for 177Lu-PSMA-617 in September 2021 based on positive data from the multi-center Phase III VISION study, which measured overall survival and progression-free survival in mCRPC patients whose disease progressed despite treatment.
177Lu-PSMA-617 is manufactured by Novartis.
Two Agents Approved For Psma
On May 26, FDA approved piflufolastat F 18 for use in a type of imaging procedure called PSMA PET in people with prostate cancer. The approval covers the use of piflufolastat F 18 in patients suspected of having metastatic prostate cancer or recurrent prostate cancer . Last year, the agency approved another imaging agent for PSMA PET, Ga 68 PSMA-11, for the same uses, but its use is largely limited to the two institutions where it is made.
In a statement, Lantheus, which manufactures piflufolastat F 18, said the imaging agent will be immediately available in parts of the mid-Atlantic and southern regions with broad availability across the U.S. anticipated by year end.
PSMA is often overproduced by prostate cancer cells but is generally not produced by most normal cells, making it an excellent target for both PET imaging and targeted systemic radiation therapy like 177Lu-PSMA-617, Dr. Morris said.
How To Get A Psma Pet Scan
- PSMA PET scans are offered at UCSF Radiology China Basin location in the San Francisco Bay area.
- For UCSF patients, please reach out to Radiology Scheduling directly to schedule your PSMA scan.
- For non-UCSF facilities referring patients that are new to UCSF, please fax the following to 353-7299 for patient registration to be completed prior to scheduling:
- Patient Exam Order
- Insurance Information and Authorization
Also Check: When Should A Man Have A Prostate Exam
Why Choose Sheba For Cancer Treatment In Israel
There are several excellent choices for cancer treatment in Israel, but The Cancer Center at Sheba Medical Center stands out for several reasons:
- Overall treatment of 50,000 cancer patients every year.
- Of those, 4,500 are new patients.
- About 35,000 patients each year are treated on an outpatient basis.
- About 37,000 patients are seen in our interdisciplinary oncology clinics every year.
- Our inpatient Oncology Department has 30 beds.
- Each year 2,000 new patients are treated in our Radiation Therapy Department.
- We provide brachytherapy, a form of internal radiation therapy, to 270 patients annually.
- We offer compassionate and effective palliative treatment.
- We possess the latest in modern research laboratories.
> > > This Simple Morning Test Will Fix Your Prostate
Another type of prostate issue is chronic prostatitis, or chronic pelvic pain syndrome. This condition causes pain in the lower back and groin area, and may cause urinary retention. Symptoms include leaking and discomfort. In severe cases, a catheter may be required to relieve the symptoms. If the problem is unresponsive to other treatments, your doctor may suggest a surgical procedure. If these do not work, your symptoms could progress and become chronic.
An acute bacterial infection can cause a burning sensation. Inflammation of the prostate can affect the bladder and result in discomfort and other symptoms. This is the most common urinary tract problem in men under 50, and the third most common in men over 65. The symptoms of acute bacterial prostatitis are similar to those of CPPS. Patients may experience a fever or chills as a result of the infection.
A bacterial infection can also lead to prostate issues. Acute bacterial infections can be hard to treat. Some men with a bacterial infection may need to take antibiotics to prevent or treat symptoms. Symptoms of the disease include fever and chills, pain in the lower back and the tip of the penis. Some men may have blood in the urine, frequent urination, and blood in the urine. If you suffer from acute bacterial prostatitis, a medical professional should be able to prescribe you the appropriate treatments to prevent the disease.
Also Check: How Is Prostate Laser Surgery Performed
What The Approval In The Us Means For Patients
Despite recent advances in nmCRPC treatment, there remains a high unmet need for new options that not only delay the time to metastases, but also limits the sometimes burdensome side effects of therapy.
CRPC is an advanced form of the disease where the cancer keeps progressing even when the amount of testosterone is reduced to very low levels in the body.2 In the U.S., over 73,000 men are estimated to have a CRPC diagnosis in 2019.3 About one-third of men with non-metastatic CRPC go on to develop metastases within two years.4
To learn more about the impact of prostate cancer on men and their loved ones, please visit: .
1 Nubeqa® tablets . Whippany, NJ: Bayer HealthCare Pharmaceuticals, 2019.
2 Luo, Jia, Beer, Tomasz, Graff, Julie. Treatment of nonmetastatic castration-resistant prostate cancer. Oncology 2016 30:336-44.
3 Bayer internal calculation based on Tesselon data .
4 National Cancer Institute. Hormone Therapy for Prostate Cancer. . Accessed July 2019.
Targeting Psma: Not Just For Imaging
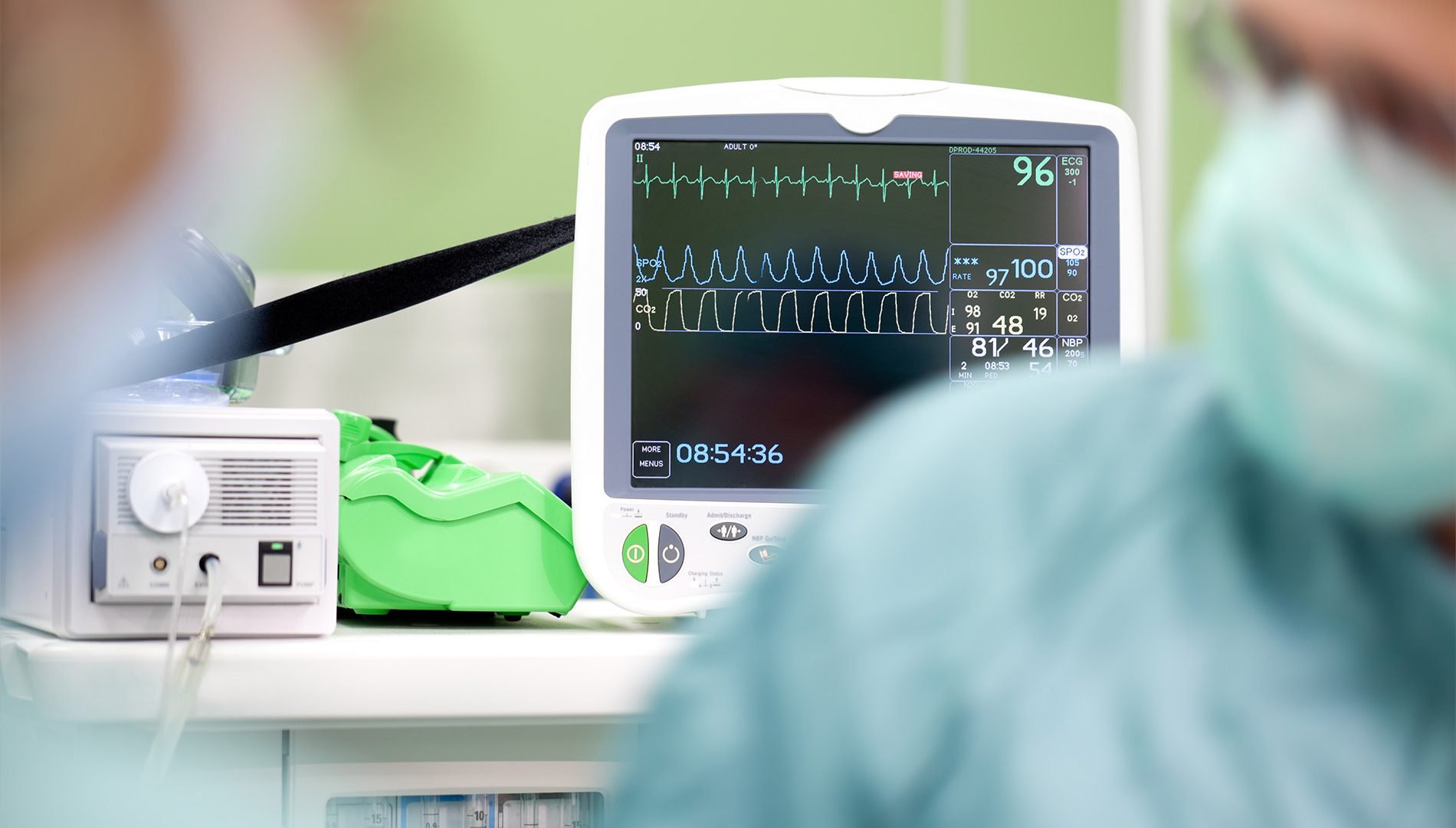
Like a number of other radiopharmaceuticals, 177Lu-PSMA-617 has two components: a drug that delivers the therapy to cancer cells and a radioactive particle. In the case of 177Lu-PSMA-617, the delivery vehicle is PSMA-617, a drug that latches onto a protein called PSMA that is often found at high levels on the surface of prostate cancer cells. The radioactive component is lutetium-177, which is being tested as a part of multiple radiopharmaceutical drugs.
As Dr. Morris explained, PSMA-617 is extremely adept at finding and locking on to the PSMA protein on cells. Once it binds to PSMA on a cancer cell, the whole molecule is internalized by the cell and the cell is exposed to a lethal dose of radiation from lutetium-177, he said.
The PSMA protein is also at the heart of a new type of imaging procedure called PSMA PET. This form of PET imaging is just starting to be used in men with prostate cancer to determine whether their cancer has spread, or metastasized, beyond the prostate. In the last several months, FDA has approved two such drugs, known as radiotracers, for PSMA PET imaging.
Recommended Reading: What Size Of Prostate Is Dangerous
Remission And The Chance Of Recurrence
A remission is when cancer cannot be detected in the body and there are no symptoms. This may also be called having no evidence of disease or NED.
A remission can be temporary or permanent. This uncertainty causes many people to worry that the cancer will come back. Although there are treatments to help prevent a recurrence, such as hormonal therapy and radiation therapy, it is important to talk with your doctor about the possibility of the cancer returning. There are tools your doctor can use, called nomograms, to estimate someone’s risk of recurrence. Understanding your risk of recurrence and the treatment options may help you feel more prepared if the cancer does return. Learn more about coping with the fear of recurrence.
In general, following surgery or radiation therapy, the PSA level in the blood usually drops. If the PSA level starts to rise again, it may be a sign that the cancer has come back. If the cancer returns after the original treatment, it is called recurrent cancer.
When this occurs, a new cycle of testing will begin again to learn as much as possible about the recurrence, including where the recurrence is located. The cancer may come back in the prostate , in the tissues or lymph nodes near the prostate , or in another part of the body, such as the bones, lungs, or liver . Sometimes the doctor cannot find a tumor even though the PSA level has increased. This is known as a PSA recurrence or biochemical recurrence.
Md Anderson Cancer Center Partnership
In furthering our goal of providing the absolute best in cancer treatment and research, Sheba Medical Center is proud to have become a MD Anderson Sister Institution in 2010. MD Anderson at the University of Texas is a leading cancer institution and was rated as the best cancer treatment center in the United States for 12 of the past 15 years by US News & World Report.
As sister institutions, Sheba and MD Anderson continue to engage in close collaboration in all aspects of oncological treatment. These aspects include education, training, development, quality assurance, nursing, research, technical support staff training, and faculty exchanges. This collaboration is completely under the auspices of Global Academic Programs, a project of MD Anderson dedicated to building a worldwide network of top cancer centers in order to effectively address and eliminate the global problem of cancer. All of the efforts and energies of Global Academic Programs are focused only on cancer diagnosis, treatment, and research.
Because of this relationship, patients afflicted with cancer have the chance to receive treatment from the absolute top cancer institutions across the globe.
You May Like: What’s The Purpose Of The Prostate
Fda Approves New Prostate Cancer Treatment
A targeted radioligand therapy, Pluvicto , has been approved for use in adults with previously treated metastatic castration-resistant prostate cancer . All eligible patients must have been previously treated with taxane-based chemotherapy and androgen receptor pathway inhibition, and test positive for a prostate-specific membrane antigen .
The efficacy of the treatment was evaluated in VISION, a randomized, open-label trial that compared best standard of care alone vs with lutetium Lu 177 vipivotide tetraxetan in 831 men with PSMA-positive mCRPC that was progressing. Patients were randomly assigned in a 2:1 ratio to receive either the best standard of care plus lutetium Lu 177 vipivotide tetraxetan, 7.4 GBq every 6 weeks until as many as 6 doses were given, or to the best standard of care alone.
VISION showed a statistically significant improvement in overall survival with lutetium Lu 177 vipivotide tetraxetan. For OS, the hazard ratio for the treatment plus best standard of care compared with best standard of care alone was 0.62 . In the treatment plus best standard of care arm, median OS was 15.3 months vs 11.3 months in the best standard of care alone arm.
The recommended dose of lutetium Lu 177 vipivotide tetraxetan is 7.4 GBq given intravenously every 6 weeks until 6 doses have been given, the cancer progresses, or unacceptable toxicity results.
Reference:
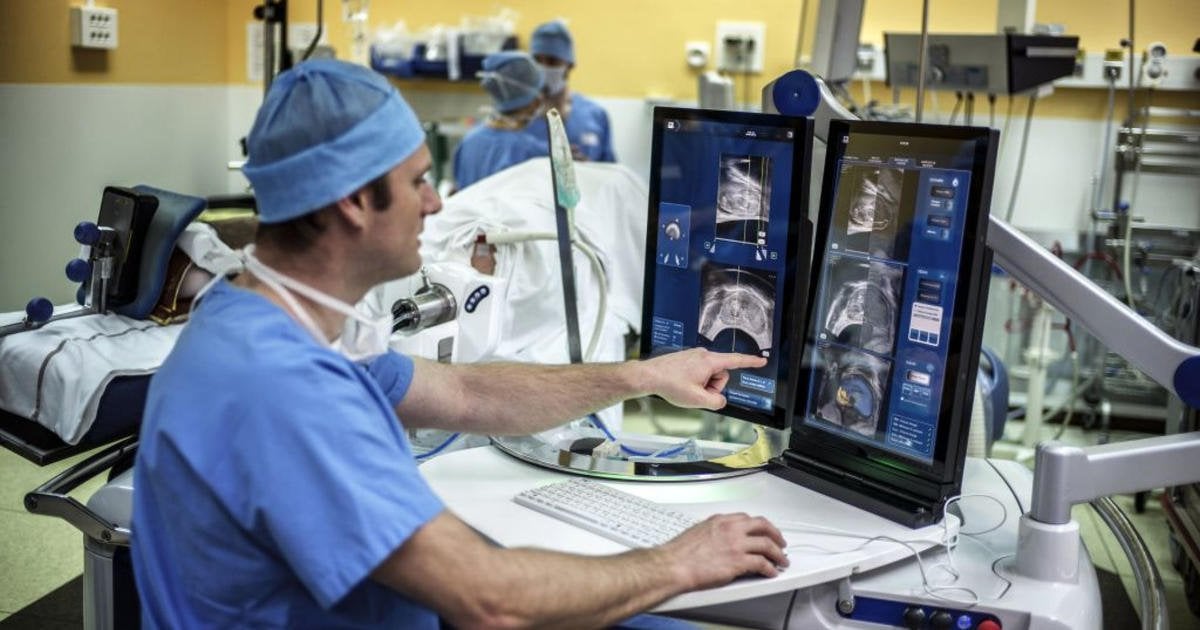
Fda Approves New Imaging Tool To Find Advanced Prostate Cancer
The Food and Drug Administration has approved a new imaging agent to detect prostate cancer after it has spread to other parts of the body.
Experts say the tracer, made by medical imaging company Lantheus, will give doctors an important visual aid to guide them to metastatic prostate cancer cells that, before now, were difficult to spot.
Prostate cancer is the second leading cause of cancer deaths in men in the United States, after lung cancer, according to the American Cancer Society. More than 34,000 men die of the disease every year.
When prostate cancer spreads, it often goes into the bones, said Dr. Michael Morris, a medical oncologist at Memorial Sloan Kettering Cancer Center in New York City. That makes it difficult to detect using traditional imaging techniques.
“It’s really hard to take pictures of what’s going on inside of bone,” Morris said, adding that traditional scans tend to find problems in the tissue surrounding bones, after damage has already been done.
“Now we don’t have to wait for that,” Morris said, who was involved with clinical trials of the tracer. “We can detect it much more clearly and much earlier than we could before.”
Also Check: Prostate Function In Human Body
How Prostate Cancer Is Treated
In cancer care, different types of doctorsincluding medical oncologists, surgeons, and radiation oncologistsoften work together to create an overall treatment plan that may combine different types of treatments to treat the cancer. This is called a multidisciplinary team. Cancer care teams include a variety of other health care professionals, such as palliative care experts, physician assistants, nurse practitioners, oncology nurses, social workers, pharmacists, counselors, dietitians, physical therapists, and others.
The common types of treatments used for prostate cancer are described below. Your care plan may also include treatment for symptoms and side effects, an important part of cancer care.
Treatment options and recommendations depend on several factors, including the type and stage of cancer, possible side effects, and the patients preferences and overall health.
Cancer treatment can affect older adults in different ways. More information on the specific effects of surgery, chemotherapy, and radiation therapy on older patients can be found another section of this website.
Because most prostate cancers are found in the early stages when they are growing slowly, you usually do not have to rush to make treatment decisions. During this time, it is important to talk with your doctor about the risks and benefits of all your treatment options and when treatment should begin. This discussion should also address the current state of the cancer:
Fda Approves Ind Application Of Accutar For Prostate Cancer Treatment Trial
The Phase I trial will assess the preliminary anti-tumour activity, safety, pharmacokinetics and tolerability of AC0176.
The US Food and Drug Administration has approved Accutar Biotechnologys Investigational New Drug application for Phase I clinical trial of AC0176 to treat metastatic castration-resistant prostate cancer .
Don’t Miss: Latest Treatment For Metastatic Prostate Cancer
Profound Trial: Most Benefit In Men With Brca2 Alterations
Olaparibs approval, announced on May 19, was based on the results of a large clinical trial called PROFOUND.
The trial enrolled men with mutations in DNA repair genes and divided them into two cohorts. Cohort A included men with alterations in the BRCA1, BRCA2, or ATM genes, each of which plays an important role in DNA repair. Cohort B included men who had alterations in a group of 12 other genes that have some involvement in repairing DNA.
All of the men in the trial had cancer that had worsened despite treatment with either abiraterone or enzalutamide , which work in different ways to block hormones in prostate cancer cells.
The 387 men in the trial were randomly assigned to either the treatment group, which received olaparib, or the control group, which received either abiraterone or enzalutamide .
In cohort A, men treated with olaparib lived more than twice as long without evidence of their cancer getting worse than men treated with abiraterone or enzalutamide: a median of 7.4 months versus 3.6 months. The treatment group in cohort A also lived longer overall, with olaparib improving survival by more than 4 months .
In addition, men treated with olaparib were far more likely to see their tumors shrink than men treated with one of the other two drugs .
In cohort B, olaparib did appear to provide some benefit in men whose tumors had alterations in some unusual genes involved in DNA repair, Dr. Sartor continued, including RAD54L and PALB2.
Sheba Ranked In The Top Tenbest Hospitals Worldwide
Sheba Medical Center sets the standard for excellence in patient-focused care. Recently, Newsweek included Sheba on their prestigious list of the Top Ten Best Hospitals Worldwide. This endorsement recognizes Sheba as a leading force in medical science and biotechnological innovation maximizing the latest technologies while always providing holistic, outstanding healthcare. Sheba is strongly committed to delivering personalized, expert medicine to everyone from all over the world.
You May Like: Seminal Vesicle Prostate Cancer Survival
Us Fda Approves Additional Indication Of Nubeqa In Combination With Docetaxel For The Treatment Of Metastatic Hormone
FOR IMMEDIATE RELEASE
Washington, D.C., Aug. 05, 2022 Bayer announced today the U.S. Food and Drug Administration has approved a supplemental New Drug Application for the oral androgen receptor inhibitor NUBEQA® with docetaxel for the treatment of adult patients with metastatic hormone-sensitive prostate cancer .1 Todays approval under the FDAs Real-Time Oncology Review pilot program was based on the pivotal Phase III ARASENS trial, which showed a significant overall survival benefit with NUBEQA plus androgen deprivation therapy and docetaxel compared to ADT and docetaxel1 NUBEQA is the only androgen receptor inhibitor approved in combination with docetaxel for mHSPC, demonstrating a 32% reduction in the risk of death compared to docetaxel alone and with a proven tolerability profile1
> > > 1 Bedtime Hack To Pee Like A Bull
An enlarged prostate can also be the cause of other problems. If the enlarged prostate is causing symptoms, the best treatment would be a natural remedy. In the meantime, there are treatments for a wide range of conditions that cause a man to experience pain. A common surgical procedure involves an electric loop, laser, or electro-stimulation. The procedure is a safe and effective option for treating enlarged or symptomatic BPH.
Also Check: Success Rate Of Prostate Cancer Treatment
What Lies Ahead: Leading The Way With Alpha Therapies
The coming years will see even more powerful forms of radioactive therapy. The MSK laboratory of radiochemist Jason Lewis and other researchers are investigating the use of alpha particles, which have a much higher energy hundreds of times more potent than the photons used in conventional radiation or beta particles. Not only do alpha particles cause more damage when they slam into cancer cells but their path of destruction is more tightly focused, sparing normal cells.
MSK is building one of the nations first dedicated alpha particle GMP labs at a U.S. academic institution.
These radiopharmaceuticals that we are creating translate very well from bench to bedside, says Dr. Lewis, Chief of the Radiochemistry and Imaging Sciences Service and Director of the Radiochemistry and Molecular Imaging Probe Core Facility. When you see these striking responses to treatment, it brings real hope for the future and our patients.
Advances in radiotheranostics are supported by The Tow Foundation, long-time contributors to MSKs mission.
- A new FDA-approved drug could be an effective treatment against prostate cancer that has spread.
- The treatment uses a molecule that seeks out and attaches to a specific protein on the cancer cell surface called PSMA
- The technology delivers radiation that damages DNA and destroys the cancer cell..