Active Surveillance And Watchful Waiting
If prostate cancer is in an early stage, is growing slowly, and treating the cancer would cause more problems than the disease itself, a doctor may recommend active surveillance or watchful waiting.
Active surveillance. Prostate cancer treatments may seriously affect a person’s quality of life. These treatments can cause side effects, such as erectile dysfunction, which is when someone is unable to get and maintain an erection, and incontinence, which is when a person cannot control their urine flow or bowel function. In addition, many prostate cancers grow slowly and cause no symptoms or problems. For this reason, many people may consider delaying cancer treatment rather than starting treatment right away. This is called active surveillance. During active surveillance, the cancer is closely monitored for signs that it is worsening. If the cancer is found to be worsening, treatment will begin.
ASCO encourages the following testing schedule for active surveillance:
-
A PSA test every 3 to 6 months
-
A DRE at least once every year
-
Another prostate biopsy within 6 to 12 months, then a biopsy at least every 2 to 5 years
Treatment should begin if the results of the tests done during active surveillance show signs of the cancer becoming more aggressive or spreading, if the cancer causes pain, or if the cancer blocks the urinary tract.
Imaging Modalities For The Diagnosis Of Castration
Beyond the current imaging modalities proposed by the RECIST guidelines , which mainly comprise CT scans and MRI, the PCWG2 also considered technetium-99 m bone scintigraphy to assess the progression of bone metastases when more than two hot spots appear however, CT scans or MRI are required when results are ambiguous .
PET-whole body CT/MRI and whole body multiparametric MRI are new imaging modalities improving the classic CT scan and MRI for the detection of new metastatic sites and the bi-dimensional assessment of progression using different radiotracers as Ga-68-PSMA-11 which is one of the most specific for prostate cancer cells .
Whole-body MRI and localised multiparametric MRI are other candidate imaging modalities for re-staging patients with CRPC . Head-to-head comparative studies between Ga-68-PSMA11-PET/CT/MRI and whole-body MRI are difficult however, it seems that the efficiency of both imaging modalities depends on the metastatic sites. In any case, both imaging modalities are more efficient than those that are currently recommended .
Thus, the current definition of CRPC based on the classic imaging modalities does not represent the true stage of the disease . Additionally, new focal treatments for metastatic CRPC based on PET imaging and adding certain conjugated drugs with biological activity against prostate cells are under development . Figure 2 gather up the proposed components of a new definition of castration resistant prostate cancer.
Ongoing Trials And Future Directions
In addition to the completed pivotal SPARTAN trial, there are a number of ongoing randomized trials testing the utility of apalutamide in a number of settings . The ACIS trial is a placebo-controlled phase III trial evaluating the utility of adding apalutamide to abiraterone/prednisone in men with mCRPC .30 The TITAN trial is assessing the utility of adding apalutamide to ADT in metastatic hormone-sensitive prostate cancer .31 The ATLAS trial is evaluating apalutamide in combination with ADT in patients with high-risk localized or locally advanced prostate cancer receiving primary radiotherapy ,32 and an Alliance Foundation trial is enrolling men with high-risk biochemically relapsed prostate cancer randomized into one of three arms to receive either degarelix, degarelix and apalutamide, or degarelix, apalutamide and abiraterone/prednisone .33
You May Like: Sleeping Pills And Enlarged Prostate
Immune Checkpoint Inhibitor Therapy
Immune checkpoint inhibitor therapy has shown clinical benefit in a number of solid tumors , but unfortunately these observations have not been replicated in patients with mCRPC . Factors such as low tumor mutational burden , loss of tumor suppressors , low prevalence of DDR genetic defects, and silencing of major histocompatibility complex-1 expression may all contribute to mCRPCs relative lack of response to ICI therapy . Two early phase-3 studies of the anti-cytotoxic T lymphocyte-associated protein-4 antibody ipilimumab both failed to meet their primary endpoint of improved OS however, recent studies investigating the efficacy of the programmed death-1 inhibitor pembrolizumab have shown promising responses in patients with mCRPC. In a single-site cohort of 48 patients with mCRPC treated with pembrolizumab, 17% had50% PSA decline with 8% having90% PSA decline as best response . These exceptional responders were found to have molecular changes , TMB-high, and mutation in LRP1b), which predispose to anti-PD-1 responses.
Completed and ongoing clinical trials investigating different ICI agents in patients with mPC have been summarized . Although monotherapy ICIs have not been successful, there are many ongoing trials to combine ICIs with standard chemotherapies or targeted therapies in order to improve clinical outcomes.
Table 3 Ongoing clinical trials investigating the administration of immune checkpoint inhibitor agents in patients with mPC
Remission And The Chance Of Recurrence
A remission is when cancer cannot be detected in the body and there are no symptoms. This may also be called having no evidence of disease or NED.
A remission can be temporary or permanent. This uncertainty causes many people to worry that the cancer will come back. Although there are treatments to help prevent a recurrence, such as hormonal therapy and radiation therapy, it is important to talk with your doctor about the possibility of the cancer returning. There are tools your doctor can use, called nomograms, to estimate someone’s risk of recurrence. Understanding your risk of recurrence and the treatment options may help you feel more prepared if the cancer does return. Learn more about coping with the fear of recurrence.
In general, following surgery or radiation therapy, the PSA level in the blood usually drops. If the PSA level starts to rise again, it may be a sign that the cancer has come back. If the cancer returns after the original treatment, it is called recurrent cancer.
When this occurs, a new cycle of testing will begin again to learn as much as possible about the recurrence, including where the recurrence is located. The cancer may come back in the prostate , in the tissues or lymph nodes near the prostate , or in another part of the body, such as the bones, lungs, or liver . Sometimes the doctor cannot find a tumor even though the PSA level has increased. This is known as a PSA-only or biochemical recurrence.
You May Like: Gleason Level 6 Prostate Cancer
If Treatment Does Not Work
Recovery from cancer is not always possible. If the cancer cannot be cured or controlled, the disease may be called advanced or terminal.
This diagnosis is stressful, and for many people, advanced cancer may be difficult to discuss. However, it is important to have open and honest conversations with your health care team to express your feelings, preferences, and concerns. The health care team has special skills, experience, and knowledge to support patients and their families and is there to help. Making sure a person is physically comfortable, free from pain, and emotionally supported is extremely important.
People who have advanced cancer and who are expected to live less than 6 months may want to consider hospice care. Hospice care is designed to provide the best possible quality of life for people who are near the end of life. You and your family are encouraged to talk with the health care team about hospice care options, which include hospice care at home, a special hospice center, or other health care locations. Nursing care and special equipment, including a hospital bed, can make staying at home a workable option for many families. Learn more about advanced cancer care planning.
After the death of a loved one, many people need support to help them cope with the loss. Learn more about grief and loss.
Survival Of Men With Metastatic Castration
There has been regular discussion here and elsewhere about the degree to which new forms of therapy have impacted patient survival since the original approval of docetaxel for treatment of metastatic, castration-resistant prostate cancer . The one thing that has always been clear is that one couldnt just add up the median survival benefits of all the new drugs and assume that the total would be the degree of absolute benefit.
We had not noticed this at the time, but as of February 2018 we have had some much more concrete information about this topic from one of the very best cancer centers in the world: the Dana-Farber Cancer Institute in Boston.
At the Genitourinary Cancers Symposium in San Francisco, earlier this year, Francini et al. reported data from a total of 583 patients, all diagnosed with and treated for mCRPC at DFCI and whose data were compiled in the DFCI Clinical Research Information System.
Francini et al. subdivided these patients into two cohorts, as follows:
Here are the core study findings:
- Average follow-up was
- 10.6 years for patients in Cohort A
- 4.6 years for patients in Cohort B
Don’t Miss: Prostate Cancer In Bones And Lungs
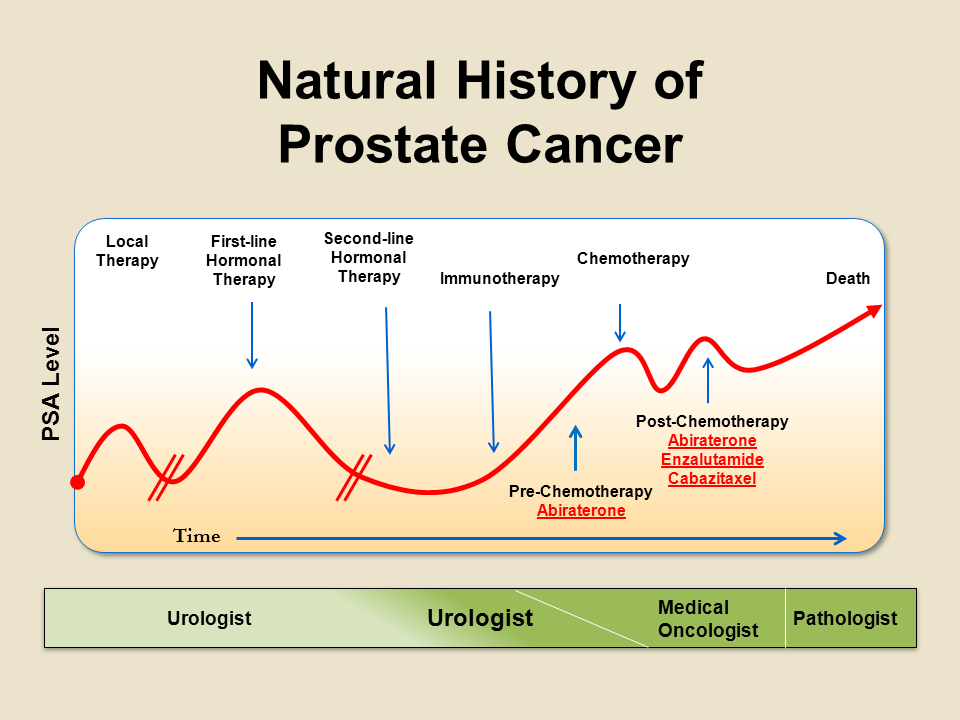
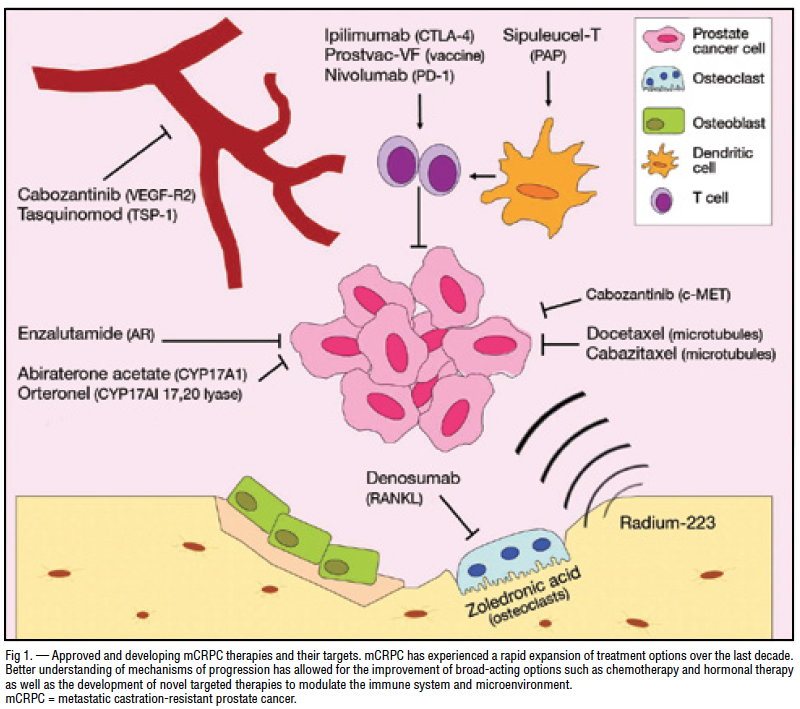
My Cancer Has Spread Outside Of My Prostate What Are My Options
If your CRPC has spread outside of your prostate and pelvis, then your treatment may be slightly different. Lab and imaging tests from your cancer specialist can determine if your cancer has spread throughout your body.
Many medications are available to treat metastatic CRPC. But specific treatment options can vary depending on where the cancer has spread. For example, a cancer specialist may choose different medications depending on if it has spread to the bones, lymph nodes, or other organs.
Regardless of where the cancer spreads, first-choice options often include:
If one or more of the above medications were unsuccessful, your cancer specialist may discuss prescribing a different one. Common alternatives include:
If you have any questions about which medication may be best for you, please speak with your cancer specialist. Most cancer types especially CRPC may be treated in several different ways.
The Pi3k/akt/mammalian Target Of Rapamycin Pathway
The PI3K pathway is one of the most critical in human cancer. Various growth factors, including insulin-like growth factor and fibroblast growth factor , regulate this pathway, leading to activation of PI3K and the formation of PIP3. PIP3 activates AKT via phosphorylation and phosphorylated Akt activates multiple molecules involved in cell survival and proliferation, including MDM2, c-myc, GSK3, nuclear factor-B and mTOR. Phosphatase and tensin homolog deleted on chromosome 10 is a lipid phosphatase that functions as the main inhibitor of PI3K/Akt signaling. Genetic alterations of the PI3K signaling pathway occur in 42% and 100% of primary and metastatic prostate cancers, respectively, suggesting this pathway is crucial in the development of CRPC.
Additional experiments showed that after 7 days of enzalutamide treatment of Ptenloxp/loxp mice, despite decreased AR transcriptional activity the tumors had not significantly regressed and were histologically similar to those before treatment, although the treatment was much more effective in transgenic mice with inducible c-myc. Further studies revealed increased Akt phosphorylation at Ser473 in the Ptenloxp/loxp mice and in LNCaP and LAPC4 AR-positive cells after castration and enzalutamide treatment, respectively. The same treatment did not increase pAkt in PC-3 cells, which are AR negative.
Also Check: What Age Does Prostate Cancer Occur
Index Patient : Asymptomatic Or Minimally Symptomatic Mcrpc Without Prior Docetaxel Chemotherapy
This patient represents a common clinical presentation seen in the CRPC setting today. These patients are characterized as having a rising PSA in the setting of castrate levels of testosterone, documented metastatic disease on radiographic imaging and no prior treatment with docetaxel chemotherapy for CRPC. The key distinction between this patient and Index Patients 3 and 4 is symptom status. Specifically, this patient is defined as having no symptoms or mild symptoms attributable to his prostate cancer. However, one must then consider whether the patient requires regular opioid pain medications for symptoms thought to be attributable to documented metastases to achieve this level of pain control. In general, if patients require regular narcotic medications for pain relief, they are not included in this category. Acknowledging these important definitions, the panel makes the following guidelines statements:
Guideline Statement 5
Clinicians should offer abiraterone plus prednisone, enzalutamide, docetaxel, or sipuleucel-T to patients with asymptomatic or minimally symptomatic mCRPC with good performance status and no prior docetaxel chemotherapy.
Discussion
Guideline Statement 6
Clinicians may offer first- generation anti-androgen therapy, ketoconazole plus steroid or observation to patients with asymptomatic or minimally symptomatic mCRPC with good performance status and no prior docetaxel chemotherapy who do not want or cannot have one of the standard therapies.
Discussion
Advances In Metastatic Castration
Matthew Fowler: Lastly, I want to focus in on treatment considerations and future directions. Is patient access a factor in considering the best treatment?
Oliver Sartor, MD: Absolutely. I hate to say it, but 1 thing were always dealing with is the ability for insurance to pay for the drugs theyre all expensive. We must work carefully with the insurance companies and work through precertifications with the insurance companies to ensure that we have the indication right for the patient. At times, we must submit the actual data, and say, This guy has a germline BRCA2 mutation. Heres the result. Here are his prior treatments. Hes already progressed on abiraterone. Hes an ideal candidate. He has metastatic castration-resistant prostate cancer . We send all that in and then work it through the precertification process. Absolutely, the reimbursement is a significant issue, and the co-pays can be problematic for patients as well. Sometimes, there are programs that can help. It depends on the individual patient, their financial resources, their insurance resources, and at times, how hard we work the system too. There are a lot of factors that go into choosing the therapy beyond simple indication.
Matthew Fowler: Given all the options now for men with metastatic CRPC, whats your preferred sequencing strategy?
Transcript edited for clarity.
You May Like: Self Check Prostate Health Test
Maintaining Quality Of Life In Patients With Nmcrpc
The patient with a rising PSA level and no radiologic evidence of metastases is usually asymptomatic from his disease but may exhibit symptoms related to age, chronic, nonmalignant comorbidities, long-term concomitant medications, adverse effects associated with ADT, and drugdrug interactions. Consequently, a consistent goal of initiating pharmacotherapy in nmCRPC should be to delay time to metastases while preserving quality of life as close to pretreatment status as possible .
Second-generation ARIs prolong survival while maintaining HRQoL in patients with nmCRPC. In clinical trials of the three approved ARIs, patient-reported outcomes captured by validated, self-administered questionnaires indicated that no treatment-induced deterioration in quality of life occurred, and that in some instances, improvement was observed in prostate cancer-specific domains .
In the SPARTAN trial, the least-squares mean change from baseline showed that HRQoL deterioration was more apparent with placebo compared with apalutamide . In PROSPER, a trend favoring enzalutamide was observed for all domains of the Functional Assessment of Cancer Therapy-Prostate, with the exception of physical well-being . In ARAMIS, time to deterioration of European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-Prostate Cancer Module outcomes demonstrated statistically and clinically significant delays with darolutamide versus placebo for urinary symptoms .
Quality Of Life With Mcrpc
According to a review published in the British Medical Journal in October 2016, you may not experience pain or other symptoms at this stage of cancer, or you may experience many. Its different for everyone. So along with treating the cancer itself, be sure to talk to your doctors about any symptoms and side effects youre experiencing in order so that the right ways to alleviate them can be found. You should also ask your care team about options for palliative care.
Because it can be very stressful to have advanced prostate cancer, and tough to talk about what it all means for your future, the ASCO urges men to have an open and honest conversation with their care team. Discuss what youre worried about, and whats important to you. There are many ways to look for and get emotional support.
Additional reporting by Andrea Peirce
Recommended Reading: Almond Milk And Prostate Cancer
Spartan Phase Iii Trial Of Apalutamide In Nmcrpc
SPARTAN was an international, randomized, double-blind, placebo-controlled phase III trial of apalutamide in patients with nmCRPC.6 The trial was conducted at 332 sites in 26 countries in North America, Europe and the Asia-Pacific region and enrolled a total of 1207 patients from October 2013 to December 2016. In order to be eligible for this trial, patients had to have histologically or cytologically confirmed adenocarcinoma of the prostate, to have castrate-resistant disease, and to have no distant metastases, as detected by a technetium-99m bone scan and computed tomography of the head, chest, abdomen and pelvis. Patients with pelvic lymph nodes measuring < 2cm in the short axis and located below the aortic bifurcation , were eligible for the trial, as were N0 patients. Eligible patients additionally were at high risk of developing metastases, defined by having a PSADT 10months while receiving continuous ADT, and were continued on ADT during the trial. Eligible patients were stratified according to PSADT , use of bone-sparing agents and nodal disease and were randomly assigned in a 2:1 ratio to receive apalutamide 240mg daily or placebo. Patients in both groups were treated on a continuous daily dosing regimen until progression, AEs, or withdrawal of consent.