Mcrpr And 177lu Psma 617 Therapy: A New Hope
Prostate cancer is the most commonly diagnosed cancer among men in the United States, apart from skin cancer. 60% of cases are diagnosed in men aged 65 years or older.
Metastatic Castration-Resistant Prostate Cancer is an advanced form of prostate cancer. Standard methods to treat prostate cancer include hormone therapy that lowers testosterone levels, known as androgen deprivation therapy .
The cancer in individuals with mCRPC does not respond to these standard therapies and continues to grow even under low testosterone conditions. The cancer then metastasizes, which means it spreads to other parts of the body, including neighbouring lymph nodes, bones, the bladder, liver, lungs, and more. For these individuals, Lu-177 may be an option.
Fda Approves Lu 177 Vipivotide Tetraxetan For Metastatic Castration
The FDA has granted approval to the agent formerly known as 177Lu-PSMA-617 for the treatment of patients with metastatic castration-resistant prostate cancer in the post androgen receptor pathway inhibition, post-taxane-based chemotherapy setting.
The FDA has granted approval to Lu 177 vipivotide tetraxetan , for the treatment of patients with metastatic castration-resistant prostate cancer in the post androgen receptor pathway inhibition, post-taxane-based chemotherapy setting, according to a press release issued by Novartis.1
“The first availability of tumor-targeted radionuclide therapy on a commercial basis will allow patients with more limited resources that might not have been able to travel for a clinical trial or overseas to receive the benefit of this treatment. The first successful phase 3 trial allows us in research to optimize the treatment, study it in earlier disease states, and explore combinations with other therapies with scientific merit. Even after this approval, I encourage clinical trial participation and/or referrals, said Scott Tagawa, MD, MS, FACP, professor of Medicine and Urology at Weil Cornell Medicine, attending physician at NewYork Presbyterin, and co-investigator of the VISION study, in an interview with Targeted OncologyTM.
“PSMA-targeted radiation is safe and leads to clinically meaningful improvements in delaying cancer progression and death in patients with few other options,” saidTagawa stated about the study.
References:
Dosimetry Of 177lu Psma Therapy
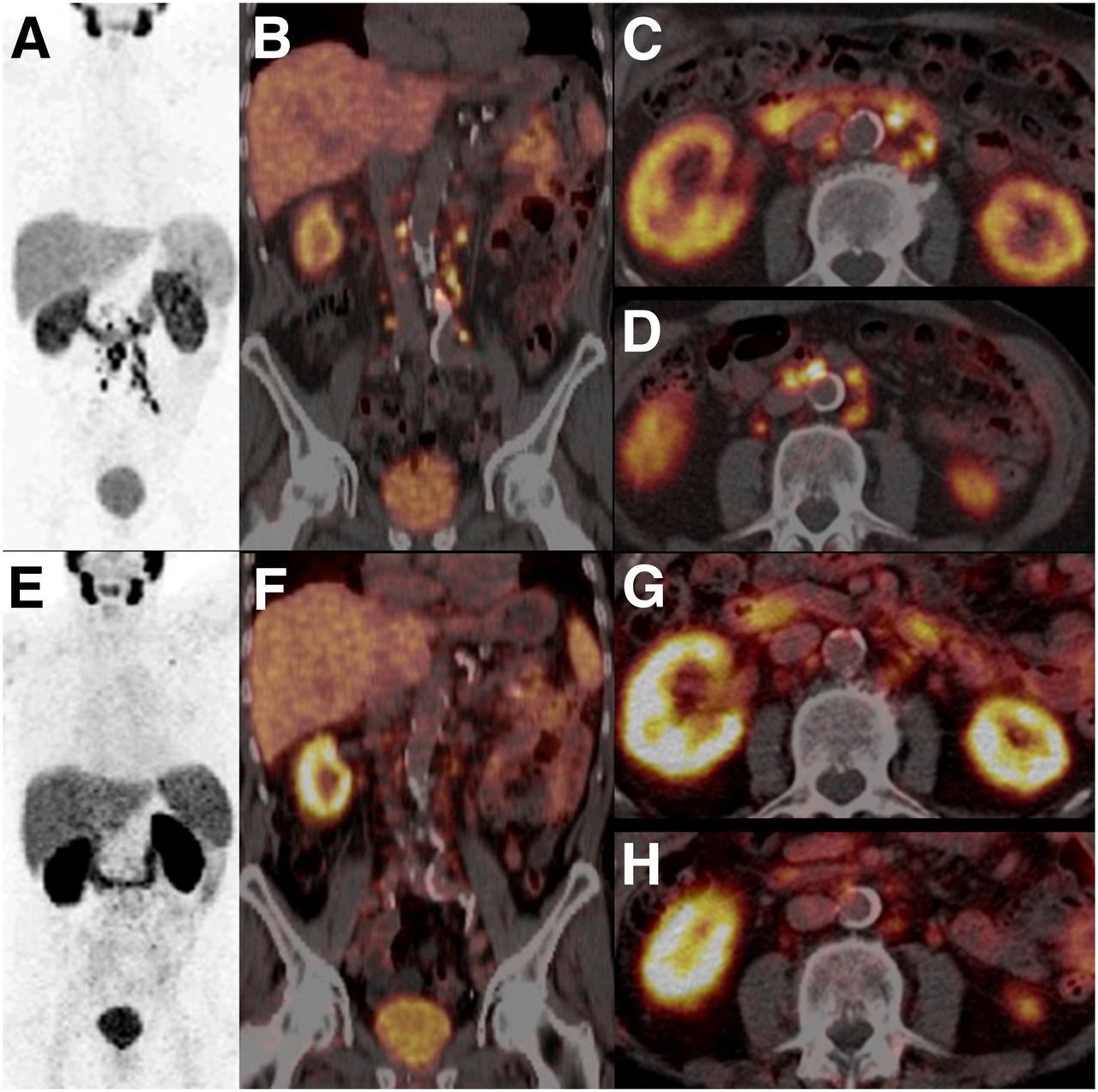
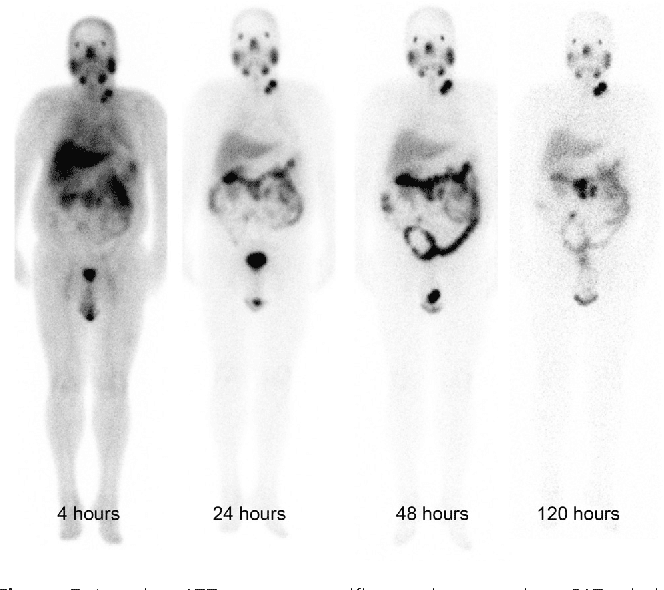
Safety and efficacy of targeted radionuclide therapies can be improved with the use of patientspecific dosimetry, which may guide successful tumour dosing and act as an early indicator of organ toxicity.32 Of particular concern in 177Lu PSMA treatment are the kidney, salivary and lacrimal glands, which suffer from radiation dosing due to the physiological behaviour of the therapeutic peptide. As a result, imagebased dosimetry at each cycle of treatment is highly recommended33.
SPECT CT whole body imaging 24 h postDose 1. 8.0 Gbq Lu PSMA Images demonstrate Lu PSMA uptake in multiple metastatic foci throughout the axial and appendicular skeleton. Serial time point imaging of the associated gamma emissions allows estimation of tumour dose delivered with each injection. SPECT CT whole body imaging 24 h postDose 2. 8.0 Gbq Lu PSMA SPECT CT images show a marked reduction in the intensity and number of Lu PSMA avid metastatic foci, which concords with the marked reduction in serum PSA in this patient.
Don’t Miss: Is Coffee Bad For Prostate
Lu 177 Therapy For Men With Prostate Cancer
- Written by Arnab Dey
Lu 177 therapy is becoming a popular choice among men suffering from prostate cancer. Over the last few years, it has been seen that in many patients, Lu-177 treatment has reduced the size of the tumor.
Men who have gone through radical prostatectomy or radiotherapy but have not seen results. They go for lu 177 therapy in Germany as their last treatment.
Yes, the therapy involves exposing your cell to beta radiation, which can damage healthy cells. However, with controlled exposure to only cancer cells, the patient can damage the prostate cancer cell and eventually kill it.
This therapy is not yet part of a clinical routine. Yet, many patients have seen positive outcomes under this treatment.
For many, Lu-177 is an unknown treatment. But, this is the reason why we are here. Today, we will discuss Lu 177 therapy and see how it works. We will also try to determine whether it is safe for your body.
Contents
The Royal Marsden To Start Offering Lutetium Psma Therapy To Treat Advanced Prostate Cancer
Patients with advanced metastatic castration resistant prostate cancer to be offered an innovative molecular therapy which precisely targets cancerous cells reducing the exposure to the rest of the body.
Lutetium-177 PSMA is an innovative therapy used to treat metastatic castration resistant prostate cancer. The therapy works to reduce the tumour size and prevents the tumour from increasing, whilst also helping to improve the symptoms that these tumours might cause.
PSMA is naturally found on the surface of prostate cells in someone with prostate cancer there is an increase of PSMA expression. If the prostate cancer has spread to other parts of the body the PSMA will also be present in those areas. When administered intravenously the Lutetium-177 PSMA ligand will travel to those areas where the PSMA is present and emits radiation that will destroy the cancer cells the treatment is targeted to the cancer with very little radiation exposure to other parts of the body.
The Royal Marsden will offer PSMA therapy, which will be administered by an expert multi-disciplinary team. Nuclear Medicine consultants, Urologist consultants, specialised Nuclear Medicine nurses, Ward nurses, Nuclear Medicine Technologists, and Nuclear Medicine Physics team will work together to ensure the best treatment and care.
All the potential side effects and the necessary precautions will be carefully explained to patients during the clinic appointment with the Nuclear Medicine consultant.
Recommended Reading: What Supplement Is Good For Prostate
Is Lutetium 177 Psma Fda Approved
177Lu-PSMA-617 is currently under review by the US FDA. In September 2021, the FDA accepted this new drug under priority review. The decision for approval is estimated to be made in the first half of 2022.
In June 2021, the FDA granted 177Lu-PSMA-617 a Breakthrough Therapy Designation . The BTD process is designed to expedite the review of drugs intended for serious conditions where early clinical results indicate improvement over already available therapies.
Note: This BTD status for 177Lu-PSMA-617 is not to be confused with the FDAs approval of another lutetium therapy: 177Lu-Dotatate, for the treatment of pancreatic and gastrointestinal cancers. While this therapy uses Lutetium-177 to treat cancers, it does not target the PSMA proteins found on prostate cancer cells.
Are You Searching For Possible Treatment Options For Metastatic Prostate Cancer
Prostate Cancer is the most common form of cancer diagnosed in men. While most patients respond well to surgery or radiation treatment, some develop advanced disease and become incurable. Prostate cancer is the second leading cause of cancer deaths in men.
The American Cancer Society estimates that over 30,000 new cases of prostate cancer were diagnosed in 2020 alone. Prostate cancer is usually treated by various methods including Surgery , radiation therapy, hormonal therapy, chemotherapy, immunotherapy as well as targeted therapy which has been recently added to the list. In the early stages of cancer, these methods might give long-term remission but in advanced metastatic prostate cancer, they only help in shrinking the tumors instead of curing the disease.
There are two main types of treatment that are commonly used in advanced metastatic castration-resistant prostate cancer i.e. chemotherapy and radiation therapy. Both methods not only kill the cancer cells but also affect the nearby healthy tissues and therefore, are not very effective against prostate cancer because they don’t target the cancer cells only.
Recommended Reading: How To Tell Prostate Cancer
Complications You Have To Know About
The implementation of therapy into clinical practice shows that no serious complications are to be feared. However, despite all efforts, the therapy can lead to complications. Since the treatment is experimental, previously unknown risks, and complications cannot be ruled out. These may include:
- Theoretical allergic reactions after drug administration, including anaphylactic shock .
- Occasionally minor hair loss in the weeks following therapy.
- Changes in taste .
- Fatigue and exhaustion .
Repeated therapy may affect renal function, so it should be checked before each course. In some cases, it may lead to a permanent loss of renal function with the need for dialysis. In addition, the continuous course of therapy may decrease saliva production and facilitate the appearance of the so-called dry mouth, which, in turn, can lead to the higher incidence of tooth decay. Temporary changes in taste may also occur as a result of treatment.
Generally, radiation is related to a risk of secondary malignancies, but such incidences with PSMA therapy are quite rare.
Is Lu 177 Therapy Safe
The radiation used in Lu-177 is designed to target only the cancer cells. With the help of theranostics, individuals get personalized treatment. This helps the experts pinpoint the cancer cells and their metastasis. During the therapy, it is ensured that the whole body is not exposed to radiation.
Before the treatment starts, blood tests will be performed to ensure your healthy tissue is not damaged. In addition, experts also perform imaging tests to ensure that the radiation is absorbed by the cancer cell correctly.
However, there are a few side effects that you might experience.
- You might feel tired for no reason.
- Your mouth will feel dry.
- Your red blood cell count might drop a little.
Also Check: Radium 223 Treatment For Prostate Cancer
How Psma Lights Up Cancer Cells
In 2021, the U.S. Food and Drug Administration issued national approval to two new prostate cancer imaging tests based on similar technology. On a PET scan, the test lights up the cancerous cells that would otherwise be hidden, enabling doctors to precisely target treatment.
Both advances in imaging and therapy rely on targeting PSMA, which is not found on most normal cells but is overexpressed in cancer cells, especially those that have spread. The PSMA molecule was cloned at MSK in the early 1990s.
The Molecular Imaging and Therapy Service, led by Heiko Schöder, played a key role in the development and testing of a slightly different PSMA-directed imaging technology at MSK.
This advance is the result of years of work by the community of physicians promoting the use of PSMA agents, Dr. Schöder says. Its gratifying to see a collaborative effort result in a breakthrough that has the potential to make a difference for so many patients with advanced prostate cancer.
Before receiving the therapy, patients in the VISION trial were scanned with PSMA-directed PET imaging to make sure enough PSMA was present in the cells to make them likely to respond to the treatment. If so, they received the radioactive drug by injection over four to six sessions, spaced six weeks apart.
As a next step, Dr. Morris and colleagues are looking into using the PSMA-directed therapy earlier rather than only after the prostate cancer has spread.
Michael Morris
How Does Lu 177 Therapy Work
PSMA is a type of protein that is expressed on the surface of the prostate cell membrane. It is believed that they have several cellular functions.
When prostate cells are healthy, they produce low levels of PSMA. However, when a person suffers from prostate cancer, the prostate cell produces high PSMA. Often 1000 times more than a normal prostate cell.
If the prostate cancer cell spreads to the other areas of the body. You will find that PSMA has also spread to those areas.
Doctors use the excessive production of the PSMA to treat cancer. As we all know that Lu-177 is a reading-based treatment. Experts use this treatment to attach a molecule to the PSMA receptor located on the cancer cell.
Lu-177 emits beta radiation that targets cancer cells and kills them.The whole process can take multiple exposures, depending on the canceled cells.
The reason why experts use PSMA receptors is that using them can be precise as the Lutetium binds itself with the PSMA. This act of targeting the cancer is often known as PRRT Peptide Receptor Radionuclide Therapy.
You May Like: Is Drinking Beer Bad For Prostate Cancer
Who Is A Candidate For Pluvicto
Men with metastatic, castrate-resistant prostate carcinoma are potential candidates for Pluvicto. In other words, these are men whose prostate cancer has progressed despite prior therapies such as surgical resection, radiation therapy, androgen receptor pathway inhibition, and/or taxane-based chemotherapy.
PET/CT imaging with PSMA is required to confirm that the metastases will accumulate PSMA, and therefore respond to Pluvicto.
Guidelines may change in the future. Anyone interested is encouraged to discuss Pluvicto with their cancer care provider. Inquiries can be directed to Alex DiFonzo at 519-3456, ext. 2351, or the theranostics team at
The Ara Theranostics Center
The Theranostics Center at ARA has been designed at our Midtown location to comfortably and safely administer the radiopharmaceuticals that are used in theranostics treatments. The treatments typically last from 2 to 6 hours depending on the radiopharmaceutical being used and the Center is made to be accommodating for patients during their stay. Wi-Fi and television are available in the infusion room. Patients may also bring a book, computer, or other quiet activity to occupy their time. Snacks and drinks are provided to patients during the treatment period. Lunch is also offered to patients whose treatments last most of the day.
Don’t Miss: Dog Prostate Cancer When To Euthanize
Why Should I Travel To Germany For Lutetium
The nuclear medicine community in Germany is extremely strong. Germany was home to the first study evaluating the use of 177Lu-PSMA-617 radioligand therapy for advanced prostate cancer patients.
At the time, the multi-center study by Rahbar et al. had the largest number of patients participating at 12 centers around Germany. The study, initiated by the German Society of Nuclear Medicine, was available online in October 2016 and published in the Journal of Nuclear Medicine in January 2017,.
Why Choose Qunomedical For Your International Medical Needs
Millions of Americans travel to other countries for their medical needs each year. There are many reasons why.
One factor is that treatment may cost less in other countries compared to the United States. This may be a more significant driving factor in recent years, as almost 5.4 million Americans lost their health insurance during the Coronavirus pandemic a record high.
Many people also seek procedures or therapies first approved in other countries before becoming available in the United States.
Planning for medical treatment abroad can be difficult. How do you know where to start? Where do you find qualified medical professionals that fit your medical needs? How do you manage hospital stays, pricing, and travel plans while you may be suffering from pain, fatigue, and other issues?
Founded by a medical professional, Qunomedical offers a concierge approach to high-quality and affordable medical treatments abroad. Every month, we connect over 6,500 patients with professionally-vetted doctors around the world. We offer 24/7 personalized support and everything you need to make an informed decision. Get your free assessment here.
Related Tags
You May Like: Refusing Treatment For Prostate Cancer
How Do I Know If Psma Therapy Is Right For Me
If you have been diagnosed with mCRPR, you may be eligible for 177Lu-PSMA-617 therapy. While the treatment is under review in the United States, it is available in countries like Germany. Find out if PSMA is suitable for you. Please reach out to our specialists at Qunomedical to discuss your unique case.
Labelling Of Lu 177 With Psma Peptides: Different Available Options
There are a number of different PSMA peptides and antibodies9, 18 that have been labelled with 177Lu, and which have been utilised in both clinical trials and for clinical use as therapy agents in men with metastatic castrateresistant prostate cancer .4, 19, 20, 21 These include the compound PSMADKFZ617, with which most patients in the published literature to date have received their therapy. This is a small molecule peptide, rather than an antibody, chemically conjugated with 177Lutetium. In an initial study in mice, it has a highly efficient internalisation into prostate cancer cells with approximately 75% of the peptide bound to the cell internalised after 3 h of incubation.22 A similar small molecule PSMA peptide ending with a different chemical conjugation also appears effective as a therapeutic agent in a number of published studies.3, 21177LuJ591, a monoclonal antibody to the PSMA receptor has been used for the therapeutic treatment of prostate cancer, with effective results, but appears to have a more limited treatment response and higher myelosuppression than seen in the studies undertaken using the smaller PSMA labelled peptides18, 23
Recommended Reading: Best Prostate Cancer Treatment Centers In The Us
When Would I Consider Using This Treatment
Lutetium-177 therapy is only used on men who suffer from cancer that has originated in the prostate and who have exhausted other previous lines of treatment. It has proven itself to be an effective therapy for managing metastatic prostate cancer or cancer of the prostate after other types of treatment have failed.
Nearly all types of prostate cancer will produce excessive PSMA, however, prior to starting the therapy, a diagnostic scan is performed to ensure the Lutetium-177 radiation will target the right areas during treatment.
How Does Lutetium Psma Therapy Work
PSMA is a type of protein found on the surface of a cell. Its located on the prostate gland, some tumours, and normal tissues.
If you have prostate cancer, youll have more PSMA than normal. If the prostate cancer has spread to other parts of the body , the PSMA will also be there.
Lutetium-177 PSMA therapy uses a molecule which attaches itself to the PSMA receptors on the cancer cells.
Before its given to you, the PSMA molecule is bound with lutetium-177. This is a radioactive substance that damages and destroys the prostate cancer cells in a targeted way.
The PSMA molecule transports the lutetium-177 direct to the tumour site. That means the rest of your body isnt exposed to radiation.
Also Check: Foods That Irritate The Prostate
Before Starting The Therapy
First off, the therapy is applicable to patients with neoplasms with enough reactivity of prostate specific membrane antigen in the cellular structure of the tumor that is no longer activated by other treatments. And therefore, before starting this specific therapy for the treatment, the PSMA levels and the indications for the PSMA technique must first be checked. This is the universal principle at European hospitals. Another obligatory condition is the patient having normal kidney and bone marrow function.
Potential causes that may be a contraindication to this technique are conditions such as:
- Severe liver and kidney dysfunction.
- Severe chronic or autoimmune diseases.
- Recent chemotherapy or bone-specific radionuclide therapy , or irradiation of a large area of the musculoskeletal system.
- Brain metastases with signs of compression.