Prevention Of Osseous Complications In Castration
3050% of patients with advanced prostate cancer suffer from pathological fractures due to bone metastases. The regular administration of a bisphosphonate or a RANK ligand inhibitor inhibits osteoclasts and reduces morbidity from bone metastases.
Zoledronic acid:
Zoledronic acid reduced skeletal complications in Phase III trials: 33% skeletal related events with zoledronic acid vs. 44% SRE in the placebo group . Serious complications include renal side effects and osteonecrosis of the jaw . Dosage: 4 mg zolendronic acid i.v. every 4 weeks. Please see section pharmacology/zoledronic acid for pharmacological details.
Denosumab:
Denosumab is a human monoclonal antibody, which interactions via RANKL and RANK inhibit osteoclasts. Denosumab has been tested in a phase III trial advantageous compared zolendronic acid: 20.7 months vs. 17.1 months until the onset of skeletal complications. Denosumab is administered subcutaneously, the dosage is 120 mg every 4 weeks. The risk of the osteonecrosis of the jaw is comparable to zoledronic acid . For the prevention of osteoporosis, the dosage of denosumab is 60 mg s.c. every 6 months. Please see section pharmacology/denosumab for pharmacological details.
If Treatment Does Not Work
Recovery from cancer is not always possible. If the cancer cannot be cured or controlled, the disease may be called advanced or terminal.
This diagnosis is stressful, and for many people, advanced cancer may be difficult to discuss. However, it is important to have open and honest conversations with your health care team to express your feelings, preferences, and concerns. The health care team has special skills, experience, and knowledge to support patients and their families and is there to help. Making sure a person is physically comfortable, free from pain, and emotionally supported is extremely important.
People who have advanced cancer and who are expected to live less than 6 months may want to consider hospice care. Hospice care is designed to provide the best possible quality of life for people who are near the end of life. You and your family are encouraged to talk with the health care team about hospice care options, which include hospice care at home, a special hospice center, or other health care locations. Nursing care and special equipment, including a hospital bed, can make staying at home a workable option for many families. Learn more about advanced cancer care planning.
After the death of a loved one, many people need support to help them cope with the loss. Learn more about grief and loss.
Future Prospects In The Treatment Of Castration
Many new agents for the treatment of castration-resistant prostate cancer are currently tested in clinical trials.
Drugs targeting androgen signal transduction:
Darolutamide prolonged time to first evidence of bone metastases in patients with M0 CRPC in the phase III ARAMIS trial approval in Europe is expected in 2020 .
PARP inhibitors:
The poly-ADP-ribose polymerase participates in DNA repair of the cell. The PARP inhibitors olaparib and rucaparib showed significant response in patients with CRPC and BRCA mutations in phase 3 studies Bono2020}, with first approvals in the U.S. .
177Lu-PSMA ligands:
Lutetium showed in uncontrolled studies a PSA response of 50\,\% in heavily pretreated patients .
Sipuleucel-T:
Sipuleucel-T is an immunotherapeutic agent to treat castration-resistant prostate carcinoma. Leukocytes from patients are obtained by plasmapheresis. Antigen-presenting cells are incubated with a specific fusion protein . The activated leukocytes are retransfused . A significantly better survival rates with low toxicity has been reported in the pivotal trial : median survival 25.8 vs. 21.7 months, three years survival rate 32% versus 23% . The drug has been approved in the USA in 2010. Criticism exists because of the high cost of Sipuleucel-T as well as Sipuleucel-T was not tested in comparison to another therapeutic agent such as docetaxel. The product was taken off the market by the company in 2015.
TAK-700:
Cabozantinib:
OGX-011:
OGX-427:
Ipilimumab:
Don’t Miss: What Kind Of Cancer Is Prostate Cancer
Immunotherapy And Prostate Cancer
Although immunotherapy has gained momentum in prostate cancer therapeutics, the vast majority of agents tested in this disease have yielded disappointing results, and in the case of anti-CTLA-4 and GVAX therapy, new concerns of safety have been raised.-
Figure 2
Overall survival in the sipuleucel-T trial. Reprinted with permission from the New England Journal of Medicine Massachusetts Medical Society. Reproduced from: Kantoff et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. Available at:
Although the immune effects of this product have been characterized in some detail, the appropriate CRPC patient who is likely to obtain the greatest benefit from treatment with sipuleucel-T requires further investigation. Current appropriate candidates for this agent include only those with asymptomatic or mildly symptomatic disease. Clinically, one difficulty with sipuleucel-T is the lack of effect on PSA, symptoms, or radiographic parameters. That is, benefit to an individual patient is difficult to gauge, and the most beneficial timing of when to initiate sipuleucel-T and any subsequent therapy is still unknown.
Despite the clinical and logistical challenges involved with these approaches, immunotherapy will likely continue playing a major role in prostate cancer therapeutics especially as newer agents are developed and the appropriate individual who can truly benefit from these interventions is defined.
Apalutamide For M0 Crpc:
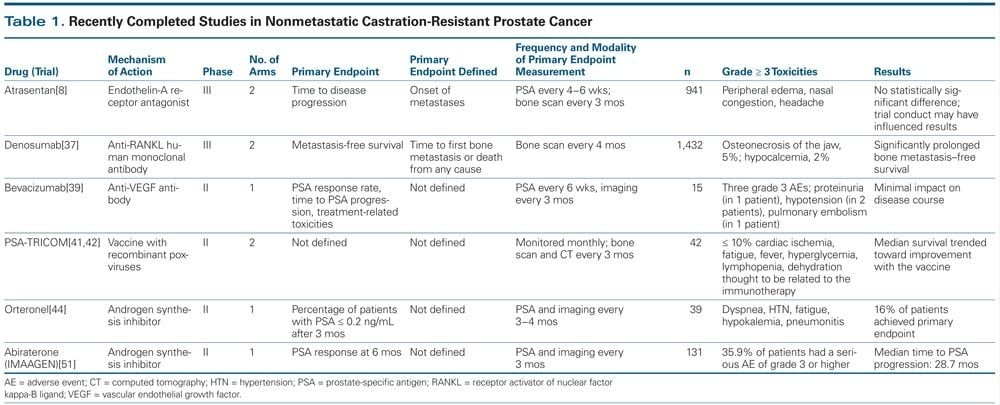
Apalutamide is an androgen receptor antagonist which prevents translocation of the activated androgen receptor into the nucleus and thus transcription of target genes. Apalutamide is approved for the treatment of high risk M0 CRPC. The dosage is 240 mg p.o. once daily in combination with standard androgen deprivation. Apalutamide was shown to prolong median metastasis-free survival in M0 CRPC with high risk . Important side effects include skin rashes see pharmacology/apalutamide for more details. Treatment is stopped if the disease shows progression or the treatment is not tolerated.
Recommended Reading: New Radiation Treatment For Prostate Cancer
Mutational And Genetic Testing In Mcrpc
All cancers, including prostate cancer, arise because of genetic mutations in cells. The first type mutations to be appreciated as causative in driving mCRPC were in genes involved in repair of damaged DNA. These mutations, often hereditary, were identified long ago as predisposing to the development of breast and ovarian cancers in women.
In prostate cancer, mutations in one of these DNA-repair genes are found mostly in late-stage prostate cancer in about 12% to 20% of patients. These mutations predict whether treatment with drugs known as PARP inhibitors might be effective. The FDA has already approved two drugs in this category: olaparib and rucaparib . New clinical guidelines now dictate testing for these mutations in tumors of mCRCP patients.
A recent study reported that men with deficiencies in one of 13 genes related to DNA damage repair had a higher response rate and a longer progression-free and overall survival when treated with the PARP inhibitor olaparib versus an anti-androgen treatment.
A new drug called berzosertib, an inhibitor of the DNA-repair protein ATR, has shown very promising results in a variety of cancers with relevant mutations, and is currently being tested in a trial for mCRPC in combination with chemotherapy.
Survival Of Men With Metastatic Castration
There has been regular discussion here and elsewhere about the degree to which new forms of therapy have impacted patient survival since the original approval of docetaxel for treatment of metastatic, castration-resistant prostate cancer . The one thing that has always been clear is that one couldnt just add up the median survival benefits of all the new drugs and assume that the total would be the degree of absolute benefit.
We had not noticed this at the time, but as of February 2018 we have had some much more concrete information about this topic from one of the very best cancer centers in the world: the Dana-Farber Cancer Institute in Boston.
At the Genitourinary Cancers Symposium in San Francisco, earlier this year, Francini et al. reported data from a total of 583 patients, all diagnosed with and treated for mCRPC at DFCI and whose data were compiled in the DFCI Clinical Research Information System.
Francini et al. subdivided these patients into two cohorts, as follows:
Here are the core study findings:
- Average follow-up was
- 10.6 years for patients in Cohort A
- 4.6 years for patients in Cohort B
Don’t Miss: Are There Any Signs Of Prostate Cancer
Imaging Modalities For The Diagnosis Of Castration
Beyond the current imaging modalities proposed by the RECIST guidelines , which mainly comprise CT scans and MRI, the PCWG2 also considered technetium-99 m bone scintigraphy to assess the progression of bone metastases when more than two hot spots appear however, CT scans or MRI are required when results are ambiguous .
PET-whole body CT/MRI and whole body multiparametric MRI are new imaging modalities improving the classic CT scan and MRI for the detection of new metastatic sites and the bi-dimensional assessment of progression using different radiotracers as Ga-68-PSMA-11 which is one of the most specific for prostate cancer cells .
Whole-body MRI and localised multiparametric MRI are other candidate imaging modalities for re-staging patients with CRPC . Head-to-head comparative studies between Ga-68-PSMA11-PET/CT/MRI and whole-body MRI are difficult however, it seems that the efficiency of both imaging modalities depends on the metastatic sites. In any case, both imaging modalities are more efficient than those that are currently recommended .
Thus, the current definition of CRPC based on the classic imaging modalities does not represent the true stage of the disease . Additionally, new focal treatments for metastatic CRPC based on PET imaging and adding certain conjugated drugs with biological activity against prostate cells are under development . Figure 2 gather up the proposed components of a new definition of castration resistant prostate cancer.
Mechanisms Of Resistance In Castration
Thenappan Chandrasekar, Joy C. Yang, Allen C. Gao, Christopher P. Evans
Department of Urology, University of California, Davis, CA, USA
Correspondence to:
Keywords: Castration-resistant disease progression drug resistance prostatic neoplasms
Submitted Dec 21, 2014. Accepted for publication Feb 28, 2015.
doi: 10.3978/j.issn.2223-4683.2015.05.02
Also Check: Is Surgery The Best Option For Prostate Cancer
Remission And The Chance Of Recurrence
A remission is when cancer cannot be detected in the body and there are no symptoms. This may also be called having no evidence of disease or NED.
A remission can be temporary or permanent. This uncertainty causes many people to worry that the cancer will come back. Although there are treatments to help prevent a recurrence, such as hormonal therapy and radiation therapy, it is important to talk with your doctor about the possibility of the cancer returning. There are tools your doctor can use, called nomograms, to estimate someone’s risk of recurrence. Understanding your risk of recurrence and the treatment options may help you feel more prepared if the cancer does return. Learn more about coping with the fear of recurrence.
In general, following surgery or radiation therapy, the PSA level in the blood usually drops. If the PSA level starts to rise again, it may be a sign that the cancer has come back. If the cancer returns after the original treatment, it is called recurrent cancer.
When this occurs, a new cycle of testing will begin again to learn as much as possible about the recurrence, including where the recurrence is located. The cancer may come back in the prostate , in the tissues or lymph nodes near the prostate , or in another part of the body, such as the bones, lungs, or liver . Sometimes the doctor cannot find a tumor even though the PSA level has increased. This is known as a PSA-only or biochemical recurrence.
New Treatments For Mcrpc
mCRPC remains a clinically challenging late-stage cancer with no curative treatment options. However, the newer hormonal drugs enzalutamide and abiraterone provide a tighter inhibition of androgen signaling, and are often used as first-line treatment in mCRPC. Docetaxel is used in patients for whom treatment with enzalutamide or abiraterone has failed, but it can also be a first-line treatment for mCRPCthis is a decision usually made by oncologists that takes into account patient-specific considerations. Bone metastases, very prevalent in prostate cancer, are treated with Radium-223 or radiation to reduce pain.
In recent years, what has changed in treatment of mCRPC is that clinical guidelines now include mutational testing and analysis of markers to predict the potential effectiveness of newer treatments that involve the immune system.
Don’t Miss: Is Painful Ejaculation A Sign Of Prostate Cancer
Importance Of The Ar In Castration
The AR is a member of the nuclear receptor superfamily that acts as a ligand-dependent transcription factor and modulates the growth and development of prostate epithelial cells. The role of AR in CRPC has been extensively studied. Despite initial data suggesting the loss of AR activity in this setting, AR overexpression in tissue specimens from castrate-resistant tumors indicates the importance of this gene even in the context of a testosterone-deprived state. Although the true biological mechanism responsible for the development of CRPC is not well understood, some of the proposed mechanisms include AR activation in a ligand-independent manner,, changes in the level of intratumoral ligands, AR gene amplification, changes in AR coregulatory molecules, and activating AR mutations., –
How Does Hormone Therapy Work Against Prostate Cancer

Early in their development, prostate cancers need androgens to grow. Hormone therapies, which are treatments that decrease androgen levels or block androgen action, can inhibit the growth of such prostate cancers, which are therefore called castration sensitive, androgen dependent, or androgen sensitive.
Most prostate cancers eventually stop responding to hormone therapy and become castration resistant. That is, they continue to grow even when androgen levels in the body are extremely low or undetectable. In the past, these tumors were also called hormone resistant, androgen independent, or hormone refractory however, these terms are rarely used now because the tumors are not truly independent of androgens for their growth. In fact, some newer hormone therapies have become available that can be used to treat tumors that have become castration resistant.
You May Like: How Severe Is Prostate Cancer
Guidelines: Prostate Cancer/management/locally Advanced And Metastatic/castration
Defining castrate-resistant prostate cancer has been a matter of much consideration due to:
- the heterogenous manifestations of prostate cancer progression, and
- the fact some patients who progress with a castrate-level of testosterone respond to second-line hormone manipulations.
Therefore a consensus statement has been developed by the Prostate Cancer Clinical Trials Working Group on what defines progression to ensure standard entry criteria onto a clinical trial. This in turn provides guidance to physicians treating patients outside a clinical trial. Castrate-resistant prostate cancer is defined as progressive disease despite castrate levels of testosterone. Progression can be deemed to have occurred based on changes in PSA and/or increase of measurable disease and/or increasing burden of disease on bone scan, while controlling for antiandrogen withdrawal responses. These criteria are standardised by assessments and include:
- Progression of measurable disease. Whether progression of measurable disease is the same as the RECIST definition . Increasing soft tissue castrate-resistant prostate cancer can occur in the absence of a rising or even detectable PSA. Only lymph nodes greater than or equal to 2cm in diameter should be used to assess changes in size.
Other sites of disease can also be evidence of prostate cancer progression, such as worsening epidural lesions. Radiographic and/or clinical documentation of disease in these sites would also qualify as progression.
Studies With The Combination Of Several Substrates For The Prevention Of Prostate Cancer
The study data is contradictory.
SELECT trial:
Substitution with selenium, vitamin E, the combination of selenium with vitamin E or placebo . No differences between treatment groups and placebo
SU.VI.MAX study:
The daily substitution with vitamin C, vitamin E, beta-carotene, selenium and zinc reduces the incidence rate of PCA : prostate cancer risk halved in patients with a PSA below 3 ng/ml. However, the PSA concentration did not change due to the substitution and is therefore not a suitable biochemical marker for future prevention trials. In patients with pathological PSA levels, the reduction of prostate cancer risk was low .
You May Like: Docetaxel Side Effects Prostate Cancer
Summary And Future Directions
With antitumor activity demonstrated across a wide mechanistic spectrum including novel hormonal maneuvers , immunotherapy, and chemotherapeutics , the management of CRPC is currently undergoing significant transformation. Manipulation of the hormonal axis remains the cornerstone of treatment, with novel adrenal androgen inhibitors demonstrating robust antitumor activity. These agents are now being tested in earlier disease stages to hopefully augment existing approaches. Immunotherapy still faces challenges with regard to exact antitumor mechanism, patient selection, and lack of intermediate clinical endpoints to inform clinical decision-making. As a result, a more precise mechanistic understanding of the antitumor effect of immunotherapy is needed to advance this approach. New chemotherapeutic approaches have demonstrated clinical benefit, but with challenges remaining regarding the management of toxicity. As is true across solid tumor oncology, robust predictive biomarkers to select specific patients for specific approaches based on anticipated benefit and/or risk is needed to further build upon the latest wave of therapeutic advances.
| Drug |
|---|
| de Bono et al | Phase 3 : C/P vs M/P in docetaxel-refractory mCRPC patients | Median OS, 15.1 vs 12.7 mo median PFS, 2.8 vs 1.4 mo ORR, 14.4% vs 4.4% in patients with measurable disease PSA decline â¥50%, 39.2% vs 17.8% | Currently FDA-approved for docetaxel-refractory mCRPC |
Quality Of Life With Mcrpc
According to a review published in the British Medical Journal in October 2016, you may not experience pain or other symptoms at this stage of cancer, or you may experience many. Its different for everyone. So along with treating the cancer itself, be sure to talk to your doctors about any symptoms and side effects youre experiencing in order so that the right ways to alleviate them can be found. You should also ask your care team about options for palliative care.
Because it can be very stressful to have advanced prostate cancer, and tough to talk about what it all means for your future, the ASCO urges men to have an open and honest conversation with their care team. Discuss what youre worried about, and whats important to you. There are many ways to look for and get emotional support.
Additional reporting by Andrea Peirce
Recommended Reading: Almond Milk And Prostate Cancer
Read Also: Psi Test For Prostate Cancer