Erleada Treatment For Castration
Having castration-resistant prostate cancer may seem daunting for everybody. When prostate cancer becomes castration-resistant it means it is not receptive to hormone therapy.
Hormone therapy for prostate cancer implies lowering testosterone levels in patients. Testosterone is the male hormone that fuels the tumors and encourages cancer growth.
When prostate cancer is castration-resistant, other treatments should be used in order to cure the disease.
Study: Erleada Demonstrates Psa Response And Increased Patient Adherence In Prostate Cancer
Real-world data show Erleada prolonged survival in both metastatic and non-metastatic castration-sensitive prostate cancer.
New data from two studies demonstrate robust prostate-specific antigen response and strong adherence rates in patients with non-metastatic castration-resistant prostate cancer treated with Janssens Erleada in the real-world clinical setting, according to data released by Janssen. A strong PSA response was also seen in a separate post-hoc analysis that showed a correlation between rapid and deep PSA response and prolonged survival in both metastatic and non-metastatic castration-sensitive prostate cancer.
These findings were presented during two sessions at the virtual American Urological Association Annual Meeting.
In metastatic prostate cancer, the percentage of patients with a PSA decline of 50 percent or 90 percent was about three times higher for patients treated with Erleada plus androgen deprivation therapy compared with patients treated with androgen deprivation therapy alone.
For non-metastatic cancer, androgen deprivation therapy showed declines in PSA, including undetectable levels in some patients.
A separate study showed that Erleada plus androgen deprivation therapy demonstrated high treatment adherence, with > 90 percent of patients adhering to therapy in both Black and non-Black subgroups.
The FDA approved Erleada in 2018 to treat patients with non-metastatic castration-resistant prostate cancer and in 2019 for metastatic disease.
National Comprehensive Cancer Network 1
Two Category 1 Preferred Recommendations
Metastatic Castration-Naive Prostate Cancer
The NCCN Clinical Practice Guidelines in Oncology include apalutamide with androgen deprivation* as a Category 1 Preferred treatment option for patients with metastatic castration-naive prostate cancer.
Non-Metastatic Castration-Resistant Prostate Cancer
The NCCN Guidelines® include apalutamide with continued androgen deprivation* as a Category 1 Preferred treatment option for patients with non-metastatic CRPC and a PSA doubling time 10 months.
*Orchiectomy, LHRH agonist, or LHRH antagonist.
The term castration-naive is used to define patients who are not on ADT at the time of progression. The NCCN Prostate Cancer Panel uses the term castration-naive even when patients have had neoadjuvant, concurrent, or adjuvant ADT as part of radiation therapy provided they have recovered testicular function.
Definition of non-metastatic : Conventional imaging studies negative for distant metastases.
Category 1:
Based upon high-level evidence, there is uniform NCCN consensus that the intervention is appropriate.
Preferred intervention:
Interventions that are based on superior efficacy, safety, and evidence, and, when appropriate, affordability.
- Metastatic castration-naive prostate cancer
- Metastatic hormone-sensitive prostate cancer
- Metastatic noncastrate prostate cancer
References
ERLEADA® is an androgen receptor inhibitor indicated for the treatment of patients with:
WARNINGS AND PRECAUTIONS
Read Also: What Size Of Prostate Requires Surgery
Erleada First Treatment For Nonmetastatic Prostate Cancer
On February 14, 2018, apalutamide was approved by the US Food and Drug Administration for the treatment of patients with nonmetastatic CRPC that is progressing despite treatment with appropriate hormone therapy.2
Apalutamide, a new-generation oral androgen receptor inhibitor, is the first nonhormone therapy drug to receive FDA approval for the treatment of nonmetastatic CRPC.2 The FDA approval of apalutamide is the first time the FDA used the clinical end point of metastasisfree survival as the basis for a drug approval. Apalutamide was granted a priority review by the FDA.
This approval is the first to use the endpoint of metastasis-free survival, measuring the length of time that tumors did not spread to other parts of the body or that death occurred after starting treatment, said Richard Pazdur, MD, Director of the FDAs Oncology Center of Excellence. In the trial supporting approval, Erleada had a robust effect on this endpoint.2
The practice guidelines from the National Comprehensive Cancer Network recently added apalutamide as part of the systemic therapy regimen for the treatment of patients with nonmetastatic CRPC.7 The NCCN guidelines recommend that the risks and benefits of apalutamide treatment should be discussed with patients, and that supportive care for bone health should be used.7
Apalutamide Data Show Correlation Of Psa And Survival In Prostate Cancer

Neeraj Agarwal, MD, discusses how the prostate-specific antigen responses correlate with survival outcomes in 2 phase 3 trials of apalutamide in patients with advanced prostate cancer.
Neeraj Agarwal, MD, a professor in the Division of Oncology, Department of Medicine, at the University of Utah School of Medicine and senior director for Clinical Research Innovation at Huntsman Cancer Institute, discusses how the prostate-specific antigen responses correlate with survival outcomes in 2 phase 3 trials of apalutamide in patients with advanced prostate cancer.
The trials evaluated for these data were the TITAN trial , which investigated patients with metastatic castration-sensitive prostate cancer, and the SPARTAN trial , which looked at patients with nonmetastatic castration-resistant prostate cancer . Both studies were double blind and placebo controlled.
Transcription:
0:08 | What we did in this analysis was to look for the degree of PSA responses and correlation of PSA responses with survival outcomes. The PSA decrease of 90%, which is a validated way to assess PSA response, so PSA 90% or undetectable PSA, which is PSA of less than 0.2 ng/mL, were considered standard measures of PSA responses in our study. Patients in both studies, in TITAN and SPARTAN, when they had these PSA responses PSA 90% or PSA of less than 0.2 ng/mL, we correlated these responses with survival outcomes.
Also Check: Prostate Cancer Erectile Dysfunction Treatment
More Common Side Effects
The more common side effects of Erleada can include*:
- fatigue
- flushing of the skin
- edema in your hands, ankles, feet, lymph nodes, or genitals
Most of these side effects may go away within a few days or a couple of weeks. If theyre more severe or dont go away, talk to your doctor or pharmacist.
* This is a partial list of the more common mild side effects from Erleada. To learn about other mild side effects, talk with your doctor or pharmacist, or visit Erleadas patient information.
Side Effects And Risks
Erleada and Xtandi work in very similar ways in the body. Therefore, they have some similar common and serious side effects.
More common side effects
These lists contain examples of more common side effects that can occur with Erleada, with Xtandi, or with both drugs .
- Can occur with Erleada:
- ischemic heart disease
Read Also: How To Reach Your Prostate
Use In Specific Populations
Men of reproductive potential should use effective contraception during apalutamide treatment and for 3 months after the last dose of apalutamide. The drug can cause harm to a fetus or loss of pregnancy. Men with a female partner with reproductive potential should use contraception. Apalutamide may increase the risk for impaired fertility in men of reproductive potential.8
No overall differences in the effectiveness of apalutamide were reported in patients aged 65 years compared with younger patients.8
What Are The Advantages Of Apalutamide
- Life expectancy: Early results suggest that apalutamide can increase life expectancy: Men who did not use apalutamide died after about 59 months on average . Men who used apalutamide died after about 66 months on average .
- Disease-related symptoms: If the prostate cancer progresses, various problems may develop or become worse, such as bone fractures or pain. Additional treatments may also become necessary, such as surgery or radiation therapy. The study suggests that apalutamide can decrease the symptoms and the number of treatments needed due to the progression of the cancer. Progressing cancer caused symptoms or made further treatment necessary in about 19 out of 100 men who took apalutamide, compared to about 25 out of 100 men in the placebo group.
- Severe kidney and urinary tract problems: Early results suggest that apalutamide has an advantage here. Kidney and urinary tract problems occurred in about 8 out of 10 men who took apalutamide, compared to about 12 out of 100 men who didn’t use apalutamide.
Also Check: What Surgery Is Done For Enlarged Prostate
The Spartan Clinical Trial
The efficacy and safety of apalutamide were evaluated in the SPARTAN trial, a double-blind, randomized, placebo-controlled, phase 3 clinical trial that included 1207 patients with nonmetastatic CRPC.5 Eligible patients were randomized to apalutamide 240 mg once daily or to placebo once daily . Patients also received a concomitant GnRH analog or had a bilateral orchiectomy.5 The primary efficacy end point in the SPARTAN study was metastasis-free survival.5
Based on results from the planned primary analysis conducted after 378 metastasis or death events were reported, the median metastasis-free survival was 40.5 months in the apalutamide group versus 16.2 months in the placebo group , a significant 24.31 months longer duration of metastasis-free survival. The time to symptomatic disease progression was also significantly longer with apalutamide versus placebo .5
The efficacy results, assessed by a blind independent review committee, are shown in the Table.5,8 These results were further supported by improvements in time to metastasis and progression-free survival achieved with apalutamide treatment versus placebo.5
Overall survival results were not mature at the time of the metastasis-free survival analysis.5
Fda Approves Erleada For Some Prostate Cancers
The US Food and Drug Administration has approved Erleada to treat men with prostate cancer that has not yet spread , but has a quickly rising PSA level while on treatment with hormone therapy, causing a big concern for cancer growth and spread. This is the first FDA-approved treatment for this high-risk type of prostate cancer, called non-metastaticcastration-resistant prostate cancer.
Erleada works by blocking the effect of androgens, a type of hormone, on the tumor. Androgens such as testosterone can help tumors grow.
The FDA based its decision on a randomized clinical trial of 1,207 men with high-risk non-metastatic, castration-resistant prostate cancer. The trial measured the amount of time patients tumors did not spread . All the men in the trial received hormone therapy, but only some also received Erleada. The men who received Erleada had no metastasis for an average 40.5 months compared to 16.2 months for the men who did not.
The FDA approved Erleada under its priority review program, which is designed to speed up approval of drugs that would significantly improve the safety or effectiveness of treating, diagnosing, or preventing a serious condition.
Common side effects of Erleada include fatigue, high blood pressure, rash, diarrhea, nausea, weight loss, joint pain, falls, hot flashes, decreased appetite, fractures and swelling in the limbs. More serious side effects include falls, fractures, and seizures.
Erleada is marketed by Janssen Pharmaceutical Companies.
Also Check: How To Get Tested For Prostate Cancer
How Long Does It Take To Work
Its not known for sure how quickly Erleada begins to work on cancer cells.
Each persons body will respond differently to Erleada. The drug may start blocking the activity of male hormones, such as testosterone, after a few days to a few weeks.
Your doctor will monitor you while take Erleada. They can give you more information about when Erleada is working for you.
Erleada For Nonmetastatic Castration
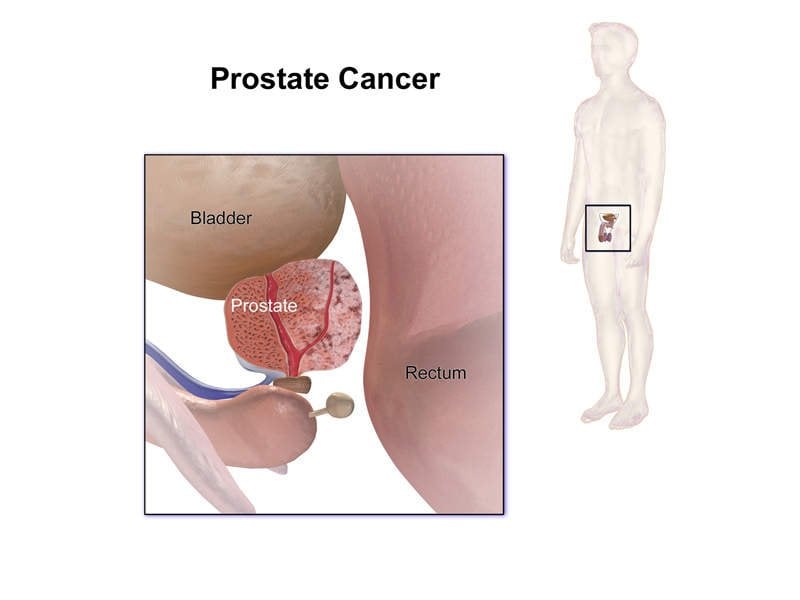
Erleada is FDA-approved to treat nonmetastatic castration-resistant prostate cancer .
Nonmetastatic prostate cancer is only found in the prostate gland. It hasnt spread to other parts of the body.
And castration-resistant prostate cancer is a type of prostate cancer that continues to grow after being treated by reducing the level of testosterone . Treatments include using certain medications or having surgery .
Effectiveness for nonmetastatic castration-resistant prostate cancer
In clinical studies, Erleada was effective in treating nmCRPC. In the studies, some people took Erleada, while other people took a placebo . Both groups of people also took a drug to help lower their testosterone level, or they had already had surgery to remove their testicles.
Researchers looked at peoples metastasis-free survival . This measurement shows how long before the mens prostate cancer spread to certain other parts of their body or they died. The study showed that:
- half of the people taking Erleada had an MFS of at least 40.5 months
- half of the people taking the placebo had an MFS of at least 16.2 months
Read Also: Ebrt Therapy For Prostate Cancer
Erleada Use With Other Drugs
In people whove had surgery to remove their testicles, Erleada can be taken alone. But in people who havent had this surgery, Erleada must be taken in combination with another type of drug.
This other type of drug is called a gonadotropin-releasing hormone analog. It belongs to a kind of treatment called analog androgen deprivation therapy .
Male hormones encourage prostate cancer cells to grow and spread. Erleada and GnRH analogs work in different ways to lower the level and effects of androgens in the body. This helps to stop prostate cancer cells from growing and spreading.
Most of the male hormones made in the body are produced in the testicles. GnRH analogs prevent the testicles from making androgens , which lowers the levels of androgens in the body.
Examples of GnRH analogs include:
- LHRH agonists, such as leuprolide or histrelin
- LHRH antagonists, such as degarelix
Small amounts of male hormones are made in other parts of the body besides the testicles and also inside cancer cells. Erleada works by blocking the effects of these hormones on prostate cancer cells. This prevents the hormones from helping the cells to grow and spread.
There arent any known interactions between Erleada and alcohol. However, drinking too much alcohol can cause many of the same side effects caused by Erleada.
Using alcohol and Erleada together can increase your risk for:
- fatigue
Erleada For Metastatic Castration
Erleada is FDA-approved to treat metastatic castration-sensitive prostate cancer .
Metastatic prostate cancer is cancer that has spread to other parts of your body.
And castration-sensitive prostate cancer is a type of prostate cancer that responds to treatment that reduces the level of testosterone in your body. Treatments include using certain medications or having surgery to remove the testicles.
Effectiveness for metastatic castration-sensitive prostate cancer
In clinical studies, Erleada was effective in treating mCSPC. In the studies, some people took Erleada, while other people took a placebo . Both groups of people also took a drug to help lower their testosterone level, or they had already had surgery to remove their testicles.
Researchers looked at peoples radiographic progression-free survival . This measurement shows how long people lived before they either had two or more new bone lesions* that showed up on a radiograph or they died.
Not everyone stayed in the study for the same amount of time. People kept taking Erleada or a placebo until their condition got worse, they had intolerable side effects, they decided to leave the study, or they died during the study. Some people stayed in the study for up to 36 weeks.
At the conclusion, the researchers found that:
* Bone lesions are areas of bone where peoples prostate cancer had spread.
You May Like: What To Eat For Prostate Health
Erleada Improves Survival Without Damaging Quality Of Life In Prostate Cancer Trial
Posted: 28 May 2021 | Hannah Balfour |
Data shows metastatic castration-sensitive prostate cancer patients treated with Erleada® were 35 percent less likely to die and had maintained quality of life.
New patient-reported outcomes data shows patients with metastatic castration-sensitive prostate cancer treated with Janssens Erleada® plus androgen deprivation therapy had a significant improvement in their overall survival, reduced the risk of death by 35 percent compared with ADT alone.
The Phase III TITAN study set out to determine if the addition of Erleada to ADT provides superior efficacy in improving radiographic progression-free survival or overall survival for participants with mHSPC. PRO data was from the pre-specified final analysis of the study, it was used to assess whether side effects from the treatment had a negative effect on patients health-related quality of life and if it worsened side effect burden, consistent with ADT alone. Erleada is an androgen receptor inhibitor.
The randomised, placebo-controlled, double-blind study enrolled 1,052 patients in 23 countries across 260 sites in North America, Latin America, South America, Europe and Asia Pacific. Patients with mCSPC were randomised 1:1 and received either Erleada plus ADT , or placebo plus ADT .
Men With Mcspc Who Received Adt Alone Were At Elevated Risk Of Disease Progression1
In men who received ADT alone for metastatic castration-sensitive prostate cancer , the median time to the development of castration resistance was < YEAR in a clinical trial*1
For men with low-volume disease2
22.7
For men with high-volume disease2
months
The median overall survival of men with mCSPC who were treated with ADT alone was YEARS in clinical trials2-6
*Time to development of castration-resistant prostate cancer was assessed in a randomized clinical trial of 790 patients with mCSPC. Time to castration-resistant prostate cancer was defined as the time until documented clinical or serologic progression with a testosterone level < 50 ng/dL .2
In this clinical trial, high-volume mCSPC was defined as the presence of visceral metastases and/or 4 bone lesions with at least 1 lesion outside of the vertebral column and/or pelvis.1
Based on patients in the control arms of 5 randomized clinical trials.2-6
ADT = androgen deprivation therapy TITAN = Targeted Investigational Treatment Analysis of Novel Antiandrogen.
References
1. Sweeney CJ, Chen YH, Carducci M, et al. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N Engl J Med. 2015 373:737-746.
2. Kyriakopoulos CE, Chen YH, Carducci MA, et al. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer: long-term survival analysis of the randomized phase III E3805 CHAARTED trial. J Clin Oncol. 2018 36:1080-1087.
Don’t Miss: Best Scan For Prostate Cancer
Erleada In Clinical Trials
A Phase 3 clinical trial called TITAN is testing Erleada plus ADT versus ADT alone in patients with mHSPC. Patients are treated until disease progression or the occurrence of treatment-related toxicity. The primary outcomes of the study are overall survival and progression-free survival.
Preliminary results from the trial, which were published in The New England Journal of Medicine, revealed that Erleada plus ADT therapy significantly improved overall survival compared to ADT alone with a 33% reduction in the risk of death. The two-year overall survival rates, after a median follow-up of 22.7 months, were 82% for Erleada plus ADT compared to 74% for ADT alone.
The safety of Erleada plus ADT was consistent with previous studies 42% of patients who received Erleada plus ADT experienced grade 3 or 4 adverse events, compared to 41% for patients receiving only ADT. The most common adverse events were high blood pressure and skin rash. Eight percent of patients in the Erleada group discontinued treatment due to adverse events, compared with 5% in the ADT-only group.
The trial is still ongoing and expected to be completed in July 2021.
Several clinical trials are recruiting prostate cancer patients now, or in the near future, to further test the effects of Erleada treatment in different populations and in combination with other therapies.