Phase 3 Study Of Immunotherapy To Treat Advanced Prostate Cancer
| The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Listing a study does not mean it has been evaluated by the U.S. Federal Government. Read our disclaimer for details. |
| First Posted : January 27, 2010Results First Posted : August 18, 2016Last Update Posted : August 18, 2016 |
| Interventional | |
| Study Design | Allocation: Randomized Intervention Model: Parallel Assignment Masking: Quadruple Primary Purpose: Treatment |
|---|---|
| Condition |
| Pre-assignment Details | 837 participants enrolled 602 randomized 598 treated. Of the 235 not randomized, 189 no longer met criteria, 28 withdrew consent, 2 suffered Adverse Events, 2 were non-compliant, 1 was lost to follow-up, and 13 were removed for other/unspecified reasons. Post-randomization, 4 no longer met criteria and were not treated |
Balancing The Scale: How Bristol Myers Squibb Is Ensuring Clinical Trial Populations Reflect Real
Guest Blog by Lorena Kuri, Director, Clinical Trial Diversity Strategy, Bristol Myers Squibb
A major news story of 2020 was the under-representation in COVID-19 clinical trials of people from Black, Latino and Native American communitiesthe ones hit hardest by the pandemic.1,2 This issue, however, isnt specific to COVID-19. Its systemic.
- Black Americans represent 12.7% of the U.S. population, but only ~4% of clinical trial populations3,4
- Hispanics and Latinos represent 18.1% of the U.S. population, but less than 8% of clinical trial populations3,5
- Native Americans represent 2% of the U.S. population, but almost 2/3 of clinical trials report having no Native American participation4,6
- Asian Americans represent almost 6% of the U.S. population, but less than 2% of clinical trial populations7
So now that the world is watching, how can we change things for the better? First, we need to think about what we can do as individuals. For me, as a Latin American woman with a platform in the healthcare industry, I am committed to advocating for diversity in clinical research to address health inequality.
Individuals, though, can only play a part in creating lasting change. Healthcare institutions must do the work of building trust and increasing access to medical research.
Breaking Down Barriers: A Case Study
Ctc Isolation Rna Extraction And Quantitative Reverse Transcriptase
Fifteen milliliters of blood was collected from each patient in EDTA vacutainer tubes . Samples were shipped in temperature-controlled packing and processed within 24 hours. The VERSA platform, as described previously,34 was used for CTC capture and extraction of mRNA.34,35 Quantitative reverse transcriptase-polymerase chain reaction was used to assess gene expression. Details are available in the Data Supplement . Expression values of CTCs isolated from 15 mL of blood were calculated as 38 â Ct values and were reported as raw data.
Also Check: What Is Malignant Neoplasm Of Prostate
Roswell Park Bristol Myers Squibb Foundation Team Up To Fight Cancer In Native And Rural Communities
Comprehensive program aims to improve access to quality care for breast, prostate cancer in high-need communities
- Initiative will mobilize 6 in-person patient navigators, 2 virtual
- Effort will address needs across state and tribes, focused on Western New York
- Key partners include Indian Health Service, federally qualified health centers
BUFFALO, N.Y. Roswell Park Comprehensive Cancer Center and the Bristol Myers Squibb Foundation announced a $3.3 million program to address the cancer burden in rural areas and Native Nations across New York State, with an emphasis on the Western New York region.
The grant from the Bristol Myers Squibb Foundation supports a service collaboration between Roswell Park, the Indian Health Service and geographically matched rural federally qualified health centers across New York. It will allow Roswell Park to provide on-site and virtual patient navigation consisting of cancer prevention, screening, treatment and education as well as available education on clinical trials, palliative care and survivorship. The primary focus will be on breast cancer and prostate cancer and secondarily on co-occurring conditions that have high rates in these communities.
Six full-time patient navigators in the high-need communities as well as two virtual navigators will be created through the grant. The funding will support:
Over the three-year project, it is expected that 3,200 community members will be engaged and educated.
Transcriptional Analysis Of Ctcs
Quantitative multiplex reverse transcriptase-polymerase chain reaction was used to measure the expression of AR, AR-Vs, AR-regulated genes, and genes upregulated in NEPC using RNA isolated from CTCs captured with an anti-EpCAM antibody. Hierarchical clustering of the transcript expression from these samples identified two distinct clusters of patients .1). Cluster 1 was characterized by low to absence of detection of AR-regulated genes. C1 included all patients with a histologic diagnosis of NEPC. By contrast, cluster 2 was enriched for patients who had high expression of transcriptional targets of AR, indicating increased AR transcriptional activity. Consistent with increased AR activity, patients classified as C2 had higher levels of serum PSA relative to the patients in cluster 1 .1). Patient and disease characteristics for each cluster are detailed in Table âTable1.1. Patient age, time since diagnosis, and percent of patients diagnosed with metastatic disease at initial diagnosis were all similar in C1 and C2. Patients in C2 were significantly more likely to have CRPC versus CSPC and bone metastases and had treatment with taxane chemotherapy.
Also Check: Prostate Cancer Marker Blood Test
Phase Ii Clinical Trials
The ENZA-CRPC trial was a phase II trial of enzalutamide in metastatic CRPC , which accrued 66 patients from 2013 to 2017. The AA-CRPC trial was a phase II trial of AA without glucocorticoid therapy for men with mCRPC,33 which accrued 58 patients between 2014 and 2016. Liquid biopsies were prospectively collected before treatment and evaluable on 21 patients from ENZA-CRPC and 27 patients from AA-CRPC, for a total of 48 patients from prospective phase II trials treated with ARSIs. These trials served as independent validation for the C2 classifier.
Bavarian Nordic Signs Major Deal For Cancer Vaccine
Bavarian Nordic, headquartered in Greater Copenhagen, and Bristol-Myers Squibb Company announced on 4 March 2015 an agreement that provides Bristol-Myers Squibb an exclusive option to license and commercialise PROSTVAC, Bavarian Nordics investigational phase 3 prostate-specific antigen-targeting cancer immunotherapy in development for the treatment of asymptomatic or minimally symptomatic metastatic castration-resistant prostate cancer .
About PROSTVACPROSTVAC is a ready to use immuno-oncology agent that stimulates an immune response that attacks prostate cancer cells. Administered subcutaneously, PROSTVAC employs Bavarian Nordics active immunotherapy technology platform. When PSA-TRICOM is presented to the immune system in the PROSTVAC regimen, cytotoxic T lymphocytes are generated that may recognise and kill PSA-expressing cancer cells and trigger an immune response to other tumor antigens.
PROSTVAC is currently being evaluated in a global, randomised, double-blind, placebo-controlled phase 3 study , that has fully enrolled at 1,298 patients with asymptomatic or minimally symptomatic metastatic castration-resistant prostate cancer .
Previous clinical studies of PROSTVAC, either as single-agent therapy or in combination with anti-androgen therapies, radiation therapies or immune checkpoint inhibitors have indicated possible therapeutic synergies for these treatment combinations and PROSTVAC has been generally well-tolerated.
Don’t Miss: What Is Prostate Specific Antigen
Study Design And Participants
CheckMate 9KD is a non-randomized, open-label, multicohort, phase 2 trial of nivolumab combined with rucaparib , docetaxel , or enzalutamide for mCRPC. Methods for the overall study and specific to cohort B have previously been described. In brief, the CheckMate 9KD study population comprises adult patients with histological confirmation of adenocarcinoma of the prostate with radiologic evidence of stage IV disease , ongoing androgen deprivation therapy or bilateral orchiectomy , and documented progressive disease per Prostate Cancer Clinical Trials Working Group 3 criteria. Eligible patients were also required to have an Eastern Cooperative Oncology Group performance status of 0 or 1 and sufficient tumor tissue obtained within 5 years before enrollment from a metastatic or primary tumor lesion not previously irradiated. Exclusion criteria included active brain metastases, conditions requiring systemic treatment with corticosteroids or other immunosuppressive medications within 14 days of start of study treatment, and prior therapy specifically targeting T-cell costimulation or immune checkpoint pathways.
Independent Validation In Prospective Trials With Aa Or Enzalutamide
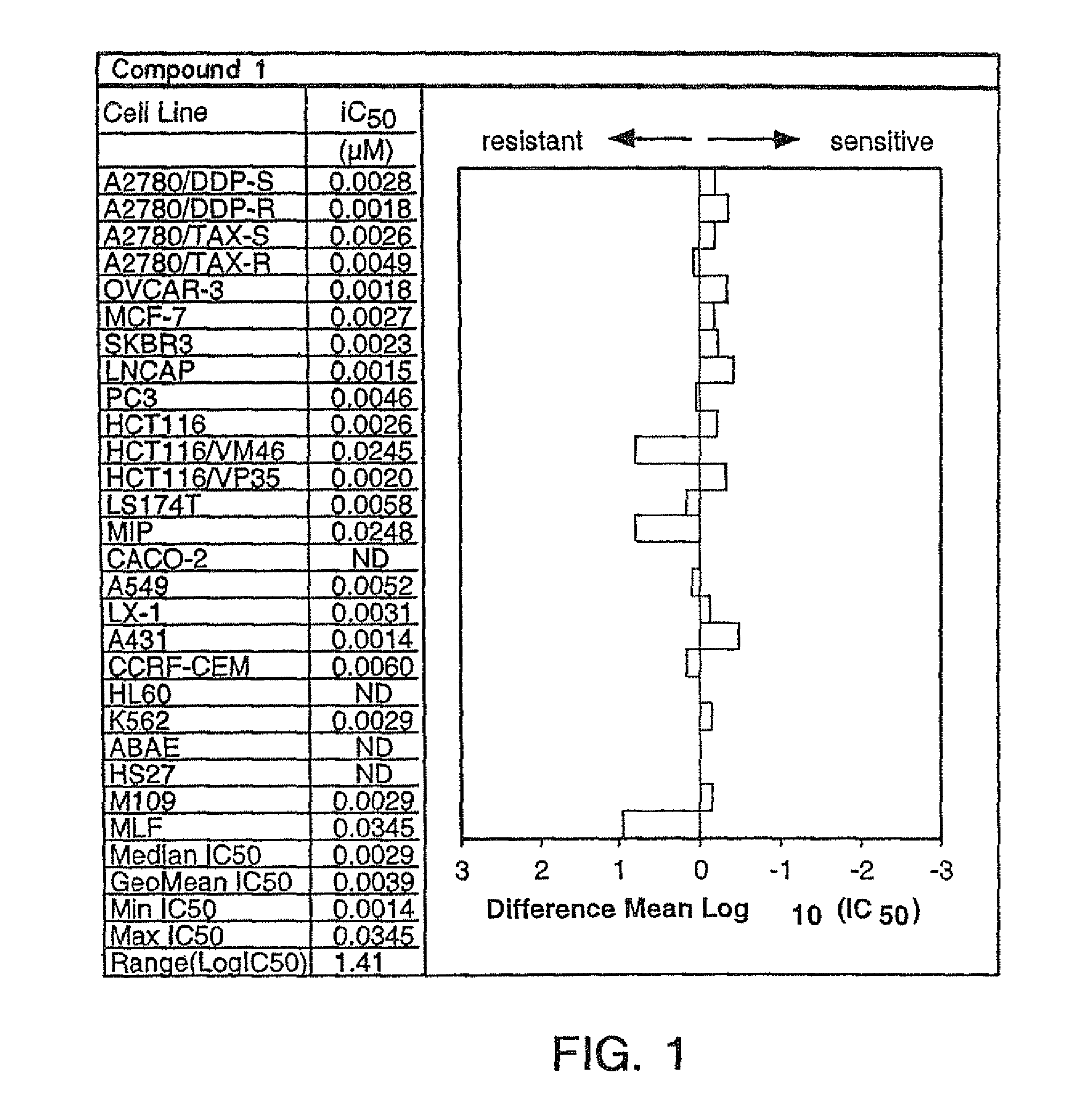
We next sought to validate our findings in two prospective phase II trials of patients with mCRPC treated with ARSIs, enzalutamide , or AA-CRPC. At time of analysis, 15 patients were deceased and the median follow-up time was 28 months in this cohort. We applied the classifier trained in UW-DFCI to these cohorts to independently identify and clinically validate C2-like patterns in CTCs. In the combined trials of ENZA-CRPC and AA-CRPC , we identified 10 patients with a C2 expression pattern. This C2 cluster was associated with worse OS 4A) in the pooled validation cohort. These independent results are consistent with the findings in our multi-institutional UW-DFCI cohort. Similarly, C2 is independently prognostic for OS relative to serum PSA, the presence of visceral metastases, age, and AR-V status using pairwise MVA in the phase II trials . Importantly, we also observed that patients in C2 had significantly shorter PSA PFS 4B) and rPFS .
C2 has worse PSA, rPFS, and OS in two multicenter phase II trials of androgen receptor signaling inhibitors. Kaplan-Meier plot of OS, PSA PFS, and rPFS for patients in C2 compared with C1 in patient treated with enzalutamide or abiraterone acetate. C1, cluster 1 C2, cluster 2 HR, hazard ratio NR, not reached OS, overall survival PFS, progression-free survival PSA, prostate-specific antigen rPFS, radiographic progression-free survival.
Recommended Reading: Can Women Have Prostate Cancer
Danish Company Could Receive Up To $975 Million In Payments
Bristol-Myers Squibb on Wednesday said it has joined with Bavarian Nordic A/S to exclusively license and commercialize the Danish companys experimental prostate-cancer treatment.
The treatment, Prostvac, is an immuno-oncology agent that stimulates the immune system to attack prostate-cancer cells.
Continue reading your article witha WSJ membership
- Copyright ©2022Dow Jones & Company, Inc. All Rights Reserved.
Copyright ©2022 Dow Jones & Company, Inc. All Rights Reserved
This copy is for your personal, non-commercial use only. Distribution and use of this material are governed byour Subscriber Agreement and by copyright law. For non-personal use or to order multiple copies, please contactDow Jones Reprints at 1-800-843-0008 or visit www.djreprints.com.
Considering A Clinical Study For Your Genitourinary Cancer
As part of the Bristol Myers Squibb commitment to patient care, we continue to explore bladder cancer, renal cell carcinoma, and prostate cancer through extensive clinical research.
You may qualify for a clinical trial.
We strongly recommend you contact BMS to report Side Effects Side Effects and other reportable events are defined hereReport Side Effects or Product Quality Complaints: Medical Information
Have questions? Live support is available 24/7 – Callor email
Learn about clinical trials and search for a clinical trial that might be right for you or a loved one.
About
You May Like: What Foods Can Prevent Prostate Cancer
Cabozantinib And Abiraterone With Checkpoint Inhibitor Immunotherapy In Metastatic Hormone Sensitive Prostate Cancer
| The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Listing a study does not mean it has been evaluated by the U.S. Federal Government.Know the risks and potential benefits of clinical studies and talk to your health care provider before participating. Read our disclaimer for details. |
| First Posted : July 20, 2020Last Update Posted : June 7, 2022 |
| Metastatic Hormone Refractory Prostate Cancer | Drug: CabozantinibDrug: NivolumabDrug: Abiraterone acetateDrug: PrednisoneProcedure: Peripheral blood collection | Phase 1 |
| Study Type : | |
| Treatment | |
| Official Title: | A Phase 1b Clinical Trial of Cabozantinib and Abiraterone With Checkpoint Inhibitor Immunotherapy in Metastatic Hormone Sensitive Prostate Cancer |
| Actual Study Start Date : |
Prospective Evaluation Of Clinical Outcomes Using A Multiplex Liquid Biopsy Targeting Diverse Resistance Mechanisms In Metastatic Prostate Cancer
Read Also: Can Men Have Their Prostate Removed
Ctc And Ctdna Changes Precede Clinical Progression
Treatment-emergent NEPC can be detected before the onset of clinical progression. Heatmap of gene expression data demonstrates the acquisition of genes commonly upregulated in NEPC as a patient is treated with androgen deprivation therapy and carboplatin and etoposide . Top panel shows ctDNA fractions at three sampled timepoints. Bottom panel shows a violin plot of the copy number profile at month 17. Blue indicates genes that pass the thresholds for identifying a copy deletion, and red indicates copy gain. Note that the violin for TP53 is distorted because of very low coverage . For PTEN and RB1, the violins have long tails, suggesting that there may be intragenic differences in copy profiles. Copy number profiles showing RB1 and PTEN exon-level data. All three patient timepoints are shown. Differences between the timepoint are a function of ctDNA fraction. Note that the high ctDNA fraction in the month 17 timepoint enables identification of an exon 1 deep deletion in PTEN and a 3â² deep deletion within RB1. AR, androgen receptor AR-V, androgen receptor splice variant ctDNA, circulating tumor DNA NA, not available NEPC, neuroendocrine prostate cancer.